Immunogenicity and Safety of a Third SARS-CoV-2 Vaccine Dose in Patients With Multiple Sclerosis and Weak Immune Response After COVID-19 Vaccination
- PMID: 35072702
- PMCID: PMC8787678
- DOI: 10.1001/jamaneurol.2021.5109
Immunogenicity and Safety of a Third SARS-CoV-2 Vaccine Dose in Patients With Multiple Sclerosis and Weak Immune Response After COVID-19 Vaccination
Abstract
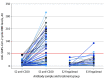
This cohort study investigates the immunogenicity and safety of a third SARS-CoV-2 vaccine dose in patients with multiple sclerosis who had a weak immune response to COVID-19 vaccination.
Conflict of interest statement
Figures
Comment in
-
Third COVID-19 jab benefits people with MS.Nat Rev Neurol. 2022 Mar;18(3):127. doi: 10.1038/s41582-022-00625-w. Nat Rev Neurol. 2022. PMID: 35121858 Free PMC article. No abstract available.
References
-
- König M, Lorentzen AR, Torgauten HM, et al. . Humoral immunity to SARS-CoV-2 mRNA vaccination in multiple sclerosis: the relevance of time since last rituximab infusion and first experience from sporadic revaccinations. J Neurol Neurosurg Psychiatry. 2021;jnnp-2021-327612. doi:10.1136/jnnp-2021-327612 - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

