Vascular smooth muscle cell-derived hydrogen sulfide promotes atherosclerotic plaque stability via TFEB (transcription factor EB)-mediated autophagy
- PMID: 35090378
- PMCID: PMC9542771
- DOI: 10.1080/15548627.2022.2026097
Vascular smooth muscle cell-derived hydrogen sulfide promotes atherosclerotic plaque stability via TFEB (transcription factor EB)-mediated autophagy
Abstract
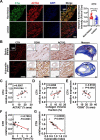
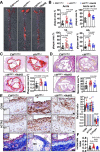
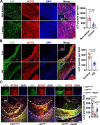
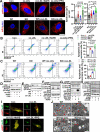
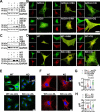
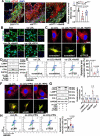
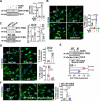
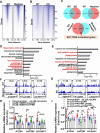
Vascular smooth muscle cells (VSMCs) contribute to plaque stability. VSMCs are also a major source of CTH (cystathionine gamma-lyase)-hydrogen sulfide (H2S), a protective gasotransmitter in atherosclerosis. However, the role of VSMC endogenous CTH-H2S in pathogenesis of plaque stability and the mechanism are unknown. In human carotid plaques, CTH expression in ACTA2+ cells was dramatically downregulated in lesion areas in comparison to non-lesion areas. Intraplaque CTH expression was positively correlated with collagen content, whereas there was a negative correlation with CD68+ and necrotic core area, resulting in a rigorous correlation with vulnerability index (r = -0.9033). Deletion of Cth in VSMCs exacerbated plaque vulnerability, and were associated with VSMC autophagy decline, all of which were rescued by H2S donor. In ox-LDL treated VSMCs, cth deletion reduced collagen and heightened apoptosis association with autophagy reduction, and vice versa. For the mechanism, CTH-H2S mediated VSMC autophagosome formation, autolysosome formation and lysosome function, in part by activation of TFEB, a master regulator for autophagy. Interference with TFEB blocked CTH-H2S effects on VSMCs collagen and apoptosis. Next, we demonstrated that CTH-H2S sulfhydrated TFEB at Cys212 site, facilitating its nuclear translocation, and then promoting transcription of its target genes such as ATG9A, LAPTM5 or LDLRAP1. Conclusively, CTH-H2S increases VSMC autophagy by sulfhydration and activation of TFEB, promotes collagen secretion and inhibits apoptosis, thereby attenuating atherogenesis and plaque vulnerability. CTH-H2S may act as a warning biomarker for vulnerable plaque.Abbreviations ATG9A: autophagy related 9A; CTH: cystathionine gamma-lyase; CQ: chloroquine; HASMCs: human aortic smooth muscle cells; H2S: hydrogen sulfide; LAMP1: lysosomal associated membrane protein 1; LAPTM5: lysosomal protein transmembrane 5; NaHS: sodium hydrosulfide hydrate; ox-LDL: oxidized-low density lipoprotein; PPG: DL- propagylglycine; TFEB: transcription factor EB; 3-MA: 3-methyladenine; VSMCs: vascular smooth muscle cells.
Keywords: Autophagy; cystathionine gamma lyase; hydrogen sulfide; plaque stability; transcription factor EB; vascular smooth muscle cell.
Conflict of interest statement
No potential conflict of interest was reported by the author(s).
Figures
References
-
- Grootaert MOJ, Moulis M, Roth L, et al. Vascular smooth muscle cell death, autophagy and senescence in atherosclerosis. Cardiovasc Res. 2018. Mar 15;114(4):622–634. - PubMed
-
- Basatemur GL, Jorgensen HF, Clarke MCH, et al. Vascular smooth muscle cells in atherosclerosis. Nat Rev Cardiol. 2019. Dec;16(12):727–744. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous
