ACE2-based capacitance sensor for rapid native SARS-CoV-2 detection in biological fluids and its correlation with real-time PCR
- PMID: 35092924
- PMCID: PMC8776344
- DOI: 10.1016/j.bios.2022.114021
ACE2-based capacitance sensor for rapid native SARS-CoV-2 detection in biological fluids and its correlation with real-time PCR
Abstract
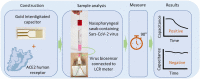
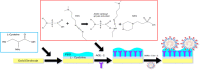
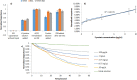
The spread of the SARS-CoV-2 and its increasing threat to human health worldwide have necessitated the development of new technological tools to combat the virus. Particular emphasis is given to the development of diagnostic methods that monitor the spread of the virus rapidly and effectively. In this study, we report the development and testing of an antibody-free biosensor, based on the immobilization of ACE2 protein on the surface of gold interdigitated electrode. When the sensor was used in laboratory conditions for targeting the virus' structural spike protein, it showed a limit of detection [LOD] of 750 pg/μL/mm2. Thereafter, the response of the sensor to swab and saliva samples from hospitalized patients was examined. The virus presence in the samples was confirmed by electrical effective capacitance measurements executed on the biosensor, and correlated with real-time PCR results. We verified that the biosensor can distinguish samples that are positive for the virus from those that are negative in a total of 7 positive and 16 negative samples. In addition, the biosensor can be used for semi-quantitative measurement, since its measurements are divided into 3 areas, the negative samples, the weakly positive and the positive samples. Reproducibility of the experiments was demonstrated with at least 3 replicates and stability was tested by keeping the sensor standby for 7 days at 4 °C before repeating the experiment. This work presents a biosensor that can be used as a fast-screening test at point of care detection of SARS-CoV-2 since it needs less than 2 min to provide results and is of simple operation.
Keywords: Antibody free; Detection; Interdigitated electrodes; SARS-CoV-2 biosensor.
Copyright © 2022 Elsevier B.V. All rights reserved.
Conflict of interest statement
The authors declare the following financial interests/personal relationships which may be considered as potential competing interests: This work has been supported by Katharsis Technologies Inc. and Galenica SA.
Figures

References
-
- Birnbaumer G.M., Lieberzeit P.A., Richter L., Schirhagl R., Milnera M., Dickert F.L., Bailey A., Ertl P. Detection of viruses with molecularly imprinted polymers integrated on a microfluidic biochip using contact-less dielectric microsensors. Lab Chip. 2009;9:3549–3556. doi: 10.1039/b914738a. - DOI - PubMed
-
- Cheng C., Cui H., Wu J., Eda S. A PCR-free point-of-care capacitive immunoassay for influenza A virus. Microchim. Acta. 2017;184:1649–1657. doi: 10.1007/s00604-017-2140-4. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

