Maternal HIV Infection as a Risk Factor for Primary Epstein-Barr Virus Infection in Kenyan Infants
- PMID: 35096607
- PMCID: PMC8790250
- DOI: 10.3389/fonc.2021.805145
Maternal HIV Infection as a Risk Factor for Primary Epstein-Barr Virus Infection in Kenyan Infants
Abstract
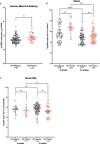
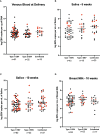
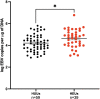
Human immunodeficiency virus (HIV) infection is known to be associated with EBV shedding in saliva suggesting an increased risk of EBV transmission to infants born to mothers with HIV at an earlier age. In this study we investigated (i) whether maternal HIV status was a risk factor for EBV in blood at delivery or for shedding in saliva and breast milk of 6- and 10-weeks post-partum mothers, (ii) if there was a difference in EBV strains shed between HIV+ and HIV- mothers, and (iii) if maternal HIV status was a determinant of EBV viral load in their infants. Samples were collected as part of a prospective cohort study that followed HIV-positive (HIV+) and HIV-negative (HIV-) pregnant women in Western Kenya through delivery and post-partum period. EBV viral load in blood was found to be significantly higher in mothers with HIV (p-value = 0.04). Additionally, a statistically significant difference was observed between EBV viral load in saliva samples and HIV status where HIV+ mothers had a higher EBV viral load in saliva at 6-weeks post-partum compared to HIV- mothers (p-value < 0.01). The difference in EBV shedding in breast milk was not found to be statistically significant. Furthermore, no difference in frequency of EBV strain was attributable to HIV- or HIV+ mothers. Interestingly, we found that infants born to HIV+ mothers had a higher EBV viral load at the time of their first EBV detection in blood than infants born to HIV- mothers and this was independent of age at detection. Overall, our study suggests that HIV infected mothers shed more virus in saliva than HIV-negative mothers and infants born to HIV+ mothers were at risk for loss of control of primary EBV infection as evidenced by higher EBV viral load following primary infection.
Keywords: Burkitt lymphoma (BL); EBV transmission; Epstein - Barr virus; HIV - human immunodeficiency virus; HIV- unexposed uninfected (HUU) infants; HIV-exposed uninfected (HEU) infants; Kenya.
Copyright © 2022 Samayoa-Reyes, Ogolla, Daud, Jackson, Sabourin, Dent and Rochford.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Piriou E, Asito AS, Sumba PO, Fiore N, Middeldorp JM, Moormann AM, et al. . Early Age at Time of Primary Epstein–Barr Virus Infection Results in Poorly Controlled Viral Infection in Infants From Western Kenya: Clues to the Etiology of Endemic Burkitt Lymphoma. J Infect Dis (2012) 205:906–13. doi: 10.1093/infdis/jir872 - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

