Evidence for a Dual-Pathway, 2-Hit Genetic Model for Focal Cortical Dysplasia and Epilepsy
- PMID: 35097204
- PMCID: PMC8789218
- DOI: 10.1212/NXG.0000000000000652
Evidence for a Dual-Pathway, 2-Hit Genetic Model for Focal Cortical Dysplasia and Epilepsy
Abstract
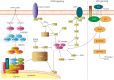
Background and objectives: The 2-hit model of genetic disease is well established in cancer, yet has only recently been reported to cause brain malformations associated with epilepsy. Pathogenic germline and somatic variants in genes in the mechanistic target of rapamycin (mTOR) pathway have been implicated in several malformations of cortical development. We investigated the 2-hit model by performing genetic analysis and searching for germline and somatic variants in genes in the mTOR and related pathways.
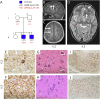
Methods: We searched for germline and somatic pathogenic variants in 2 brothers with drug-resistant focal epilepsy and surgically resected focal cortical dysplasia (FCD) type IIA. Exome sequencing was performed on blood- and brain-derived DNA to identify pathogenic variants, which were validated by droplet digital PCR. In vitro functional assays of a somatic variant were performed.
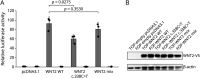
Results: Exome analysis revealed a novel, maternally inherited, germline pathogenic truncation variant (c.48delG; p.Ser17Alafs*70) in NPRL3 in both brothers. NPRL3 is a known FCD gene that encodes a negative regulator of the mTOR pathway. Somatic variant calling in brain-derived DNA from both brothers revealed a low allele fraction somatic variant (c.338C>T; p.Ala113Val) in the WNT2 gene in 1 brother, confirmed by droplet digital PCR. In vitro functional studies suggested a loss of WNT2 function as a consequence of this variant. A second somatic variant has not yet been found in the other brother.
Discussion: We identify a pathogenic germline mTOR pathway variant (NPRL3) and a somatic variant (WNT2) in the intersecting WNT signaling pathway, potentially implicating the WNT2 gene in FCD and supporting a dual-pathway 2-hit model. If confirmed in other cases, this would extend the 2-hit model to pathogenic variants in different genes in critical, intersecting pathways in a malformation of cortical development. Detection of low allele fraction somatic second hits is challenging but promises to unravel the molecular architecture of FCDs.
Copyright © 2022 The Author(s). Published by Wolters Kluwer Health, Inc. on behalf of the American Academy of Neurology.
Figures

Similar articles
-
Identification of genetic characteristics in pediatric epilepsy with focal cortical dysplasia type 2 using deep whole-exome sequencing.Mol Genet Genomic Med. 2022 Dec;10(12):e2086. doi: 10.1002/mgg3.2086. Epub 2022 Nov 7. Mol Genet Genomic Med. 2022. PMID: 36342087 Free PMC article.
-
mTOR Pathway Somatic Pathogenic Variants in Focal Malformations of Cortical Development: Novel Variants, Topographic Mapping, and Clinical Outcomes.Neurol Genet. 2023 Oct 26;9(6):e200103. doi: 10.1212/NXG.0000000000200103. eCollection 2023 Dec. Neurol Genet. 2023. PMID: 37900581 Free PMC article.
-
Somatic variants in diverse genes leads to a spectrum of focal cortical malformations.Brain. 2022 Aug 27;145(8):2704-2720. doi: 10.1093/brain/awac117. Brain. 2022. PMID: 35441233 Free PMC article.
-
Genomic and Epigenetic Advances in Focal Cortical Dysplasia Types I and II: A Scoping Review.Front Neurosci. 2021 Jan 22;14:580357. doi: 10.3389/fnins.2020.580357. eCollection 2020. Front Neurosci. 2021. PMID: 33551717 Free PMC article.
-
Epilepsy in the mTORopathies: opportunities for precision medicine.Brain Commun. 2021 Sep 25;3(4):fcab222. doi: 10.1093/braincomms/fcab222. eCollection 2021. Brain Commun. 2021. PMID: 34632383 Free PMC article. Review.
Cited by
-
Phenotypic and genotypic characterization of NPRL3-related epilepsy: Two case reports and literature review.Epilepsia Open. 2024 Feb;9(1):33-40. doi: 10.1002/epi4.12856. Epub 2023 Nov 28. Epilepsia Open. 2024. PMID: 37902097 Free PMC article. Review.
-
Identification of genetic characteristics in pediatric epilepsy with focal cortical dysplasia type 2 using deep whole-exome sequencing.Mol Genet Genomic Med. 2022 Dec;10(12):e2086. doi: 10.1002/mgg3.2086. Epub 2022 Nov 7. Mol Genet Genomic Med. 2022. PMID: 36342087 Free PMC article.
-
Identification of two rare NPRL3 variants in two Chinese families with familial focal epilepsy with variable foci 3: NGS analysis with literature review.Front Genet. 2023 Jan 6;13:1054567. doi: 10.3389/fgene.2022.1054567. eCollection 2022. Front Genet. 2023. PMID: 36685832 Free PMC article.
-
The Power of Clinical Diagnosis for Deciphering Complex Genetic Mechanisms in Rare Diseases.Genes (Basel). 2023 Jan 12;14(1):196. doi: 10.3390/genes14010196. Genes (Basel). 2023. PMID: 36672937 Free PMC article. Review.
-
Refining the electroclinical spectrum of NPRL3-related epilepsy: A novel multiplex family and literature review.Epilepsia Open. 2023 Dec;8(4):1314-1330. doi: 10.1002/epi4.12798. Epub 2023 Sep 1. Epilepsia Open. 2023. PMID: 37491868 Free PMC article. Review.
References
-
- Iffland PH II, Crino PB. Focal cortical dysplasia: gene mutations, cell signaling, and therapeutic implications. Annu Rev Pathol. 2017;12:547-571. - PubMed
-
- Marsan E, Baulac S. Review: mechanistic target of rapamycin (mTOR) pathway, focal cortical dysplasia and epilepsy. Neuropathol Appl Neurobiol. 2018;44(1):6-17. - PubMed
-
- Lim JS, Kim WI, Kang HC, et al. . Brain somatic mutations in MTOR cause focal cortical dysplasia type II leading to intractable epilepsy. Nat Med. 2015;21(4):395-400. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
