Size-Dependent Order-Disorder Crossover in Hydrophobic Hydration: Comparison between Spherical Solutes and Linear Alcohols
- PMID: 35097265
- PMCID: PMC8793046
- DOI: 10.1021/acsomega.1c05064
Size-Dependent Order-Disorder Crossover in Hydrophobic Hydration: Comparison between Spherical Solutes and Linear Alcohols
Abstract
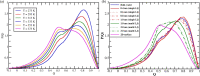
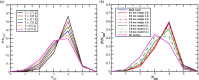
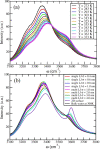
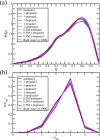
Theory and computer simulation studies have predicted that water molecules around hydrophobic molecules should undergo an order-disorder transition with increasing solute size around a 1 nm length scale. Some theories predict the formation of a clathrate-like ordered structure around smaller hydrophobic solutes (<1 nm) and the formation of disordered vapor-liquid interfaces around larger solutes (>1 nm) and surfaces. Experimental validation of these predictions has often been elusive and contradictory. High-resolution Raman spectroscopy has detected that water around small hydrophobic solutes shows a signature similar to that of bulk water at lower temperature (increased ordering and a stronger hydrogen-bonded network). Similarly, water around larger solutes shows an increasing population of dangling OH bonds very similar to higher temperature bulk water. Thus, the solute size dependence of the structure and dynamics of water around hydrophobic molecules seems to have an analogy with the temperature dependence in bulk water. In this work, using atomistic classical molecular dynamics (MD) simulations, we have systematically investigated this aspect and characterized this interesting analogy. Structural order parameters including the tetrahedral order parameter (Q), hydrogen bond distribution, and vibrational power spectrum highlight this similarity. However, in contrast to the experimental observations, we do not observe any length-dependent crossover for linear hydrophobic alcohols (n-alkanols) using classical MD simulations. This is in agreement with earlier findings that linear alkane chains do not demonstrate the length-dependent order-disorder transition due to the presence of a sub-nanometer length scale along the cross section of the chain. Moreover, the collapsed state of linear hydrocarbon chains is not significantly populated for smaller chains (number of carbons below 20). In the context of our computational results, we raise several pertinent questions related to the sensitivity of various structural and dynamical parameters toward capturing these complex phenomena of hydrophobic hydration.
© 2022 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
Similar articles
-
Structural Order of Water Molecules around Hydrophobic Solutes: Length-Scale Dependence and Solute-Solvent Coupling.J Phys Chem B. 2015 Aug 27;119(34):11346-57. doi: 10.1021/acs.jpcb.5b03449. Epub 2015 Jun 18. J Phys Chem B. 2015. PMID: 26039676
-
Water structural transformation at molecular hydrophobic interfaces.Nature. 2012 Nov 22;491(7425):582-5. doi: 10.1038/nature11570. Nature. 2012. PMID: 23172216
-
Temperature-Dependent Hydrophobic Crossover Length Scale and Water Tetrahedral Order.J Phys Chem Lett. 2018 Mar 1;9(5):1012-1017. doi: 10.1021/acs.jpclett.7b03431. Epub 2018 Feb 13. J Phys Chem Lett. 2018. PMID: 29420897
-
Recent developments in the theoretical, simulational, and experimental studies of the role of water hydrogen bonding in hydrophobic phenomena.Adv Colloid Interface Sci. 2016 Sep;235:23-45. doi: 10.1016/j.cis.2016.05.006. Epub 2016 May 18. Adv Colloid Interface Sci. 2016. PMID: 27312562 Review.
-
Structural and thermodynamic aspects of the hydrophobic effect.Adv Biophys. 1993;29:1-54. doi: 10.1016/0065-227x(93)90004-o. Adv Biophys. 1993. PMID: 8140943 Review.
Cited by
-
Spectroscopic Fingerprints of Cavity Formation and Solute Insertion as a Measure of Hydration Entropic Loss and Enthalpic Gain.Angew Chem Int Ed Engl. 2022 Jul 18;61(29):e202203893. doi: 10.1002/anie.202203893. Epub 2022 Jun 1. Angew Chem Int Ed Engl. 2022. PMID: 35500074 Free PMC article.
-
Extension of the molar absorption coefficient for non-ideal mixtures: an application to aqueous monovalent alcohol solutions.Anal Sci. 2025 Apr;41(4):353-363. doi: 10.1007/s44211-024-00703-7. Epub 2024 Dec 13. Anal Sci. 2025. PMID: 39671140
References
-
- Bagchi B.Water in Biological and Chemical Processes; Cambridge University Press: Cambridge, 2013.
LinkOut - more resources
Full Text Sources

