The tomato OST1-VOZ1 module regulates drought-mediated flowering
- PMID: 35099557
- PMCID: PMC9048945
- DOI: 10.1093/plcell/koac026
The tomato OST1-VOZ1 module regulates drought-mediated flowering
Abstract
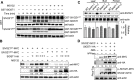
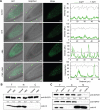
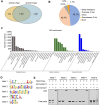
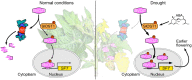
Flowering is a critical agricultural trait that substantially affects tomato fruit yield. Although drought stress influences flowering time, the molecular mechanism underlying drought-regulated flowering in tomato remains elusive. In this study, we demonstrated that loss of function of tomato OPEN STOMATA 1 (SlOST1), a protein kinase essential for abscisic acid (ABA) signaling and abiotic stress responses, lowers the tolerance of tomato plants to drought stress. slost1 mutants also exhibited a late flowering phenotype under both normal and drought stress conditions. We also established that SlOST1 directly interacts with and phosphorylates the NAC (NAM, ATAF and CUC)-type transcription factor VASCULAR PLANT ONE-ZINC FINGER 1 (SlVOZ1), at residue serine 67, thereby enhancing its stability and nuclear translocation in an ABA-dependent manner. Moreover, we uncovered several SlVOZ1 binding motifs from DNA affinity purification sequencing analyses and revealed that SlVOZ1 can directly bind to the promoter of the major flowering-integrator gene SINGLE FLOWER TRUSS to promote tomato flowering transition in response to drought. Collectively, our data uncover the essential role of the SlOST1-SlVOZ1 module in regulating flowering in response to drought stress in tomato and offer insights into a novel strategy to balance drought stress response and flowering.
© American Society of Plant Biologists 2022. All rights reserved. For permissions, please email: journals.permissions@oup.com.
Figures





Comment in
-
Escaping the drought: the OST1-VOZ1 module regulates early flowering in tomato.Plant Cell. 2022 Apr 26;34(5):1886-1887. doi: 10.1093/plcell/koac042. Plant Cell. 2022. PMID: 35157084 Free PMC article. No abstract available.
Similar articles
-
Beyond stress response: OST1 opening doors for plants to grow.Stress Biol. 2022 Oct 25;2(1):44. doi: 10.1007/s44154-022-00069-8. Stress Biol. 2022. PMID: 37676544 Free PMC article. Review.
-
Functional analysis of open stomata 1-slow anion channel associated 1-6 protein module in enhancing drought tolerance in tomato through stomatal regulation mechanisms.Int J Biol Macromol. 2025 May;308(Pt 3):142591. doi: 10.1016/j.ijbiomac.2025.142591. Epub 2025 Mar 27. Int J Biol Macromol. 2025. PMID: 40157666
-
The abiotic stress-responsive NAC-type transcription factor SlNAC4 regulates salt and drought tolerance and stress-related genes in tomato (Solanum lycopersicum).Plant Cell Rep. 2014 Nov;33(11):1851-63. doi: 10.1007/s00299-014-1662-z. Epub 2014 Jul 26. Plant Cell Rep. 2014. PMID: 25063324
-
Silencing of SlMYB55 affects plant flowering and enhances tolerance to drought and salt stress in tomato.Plant Sci. 2022 Mar;316:111166. doi: 10.1016/j.plantsci.2021.111166. Epub 2021 Dec 25. Plant Sci. 2022. PMID: 35151450
-
The interplay between ABA/ethylene and NAC TFs in tomato fruit ripening: a review.Plant Mol Biol. 2021 Jun;106(3):223-238. doi: 10.1007/s11103-021-01128-w. Epub 2021 Feb 25. Plant Mol Biol. 2021. PMID: 33634368 Review.
Cited by
-
Ubiquitylation of PHYTOSULFOKINE RECEPTOR 1 modulates the defense response in tomato.Plant Physiol. 2023 Jul 3;192(3):2507-2522. doi: 10.1093/plphys/kiad188. Plant Physiol. 2023. PMID: 36946197 Free PMC article.
-
Cloning and functional identification of apple LATERAL ORGAN BOUNDARY DOMAIN 3 (LBD3) transcription factor in the regulation of drought and salt stress.Planta. 2024 Apr 18;259(6):125. doi: 10.1007/s00425-024-04373-7. Planta. 2024. PMID: 38634979
-
Dissection of major QTLs and candidate genes for seedling stage salt/drought tolerance in tomato.BMC Genomics. 2024 Dec 3;25(1):1170. doi: 10.1186/s12864-024-11101-8. BMC Genomics. 2024. PMID: 39627739 Free PMC article.
-
ZmELF6-ZmPRR37 module regulates maize flowering and salt response.Plant Biotechnol J. 2024 Apr;22(4):929-945. doi: 10.1111/pbi.14236. Epub 2023 Nov 27. Plant Biotechnol J. 2024. PMID: 38009862 Free PMC article.
-
Beyond stress response: OST1 opening doors for plants to grow.Stress Biol. 2022 Oct 25;2(1):44. doi: 10.1007/s44154-022-00069-8. Stress Biol. 2022. PMID: 37676544 Free PMC article. Review.
References
-
- Assmann SM (2003) Open Stomata1 opens the door to ABA signaling in Arabidopsis guard cells. Trends Plant Sci 8: 151–153 - PubMed
-
- Bao F, Huang X, Zhu C, Zhang X, Li X, Yang S (2014) Arabidopsis HSP90 protein modulates RPP4-mediated temperature-dependent cell death and defense responses. New Phytol 202: 1320–1334 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

