Pyrazinamide Susceptibility Is Driven by Activation of the SigE-Dependent Cell Envelope Stress Response in Mycobacterium tuberculosis
- PMID: 35100871
- PMCID: PMC8805019
- DOI: 10.1128/mbio.00439-21
Pyrazinamide Susceptibility Is Driven by Activation of the SigE-Dependent Cell Envelope Stress Response in Mycobacterium tuberculosis
Abstract
Pyrazinamide (PZA) plays a crucial role in first-line tuberculosis drug therapy. Unlike other antimicrobial agents, PZA is active against Mycobacterium tuberculosis only at low pH. The basis for this conditional drug susceptibility remains undefined. In this study, we utilized a genome-wide approach to interrogate potentiation of PZA action. We found that mutations in numerous genes involved in central metabolism as well as cell envelope maintenance and stress response are associated with PZA resistance. Further, we demonstrate that constitutive activation of the cell envelope stress response can drive PZA susceptibility independent of environmental pH. Consequently, exposure to peptidoglycan synthesis inhibitors, such as beta-lactams and d-cycloserine, potentiate PZA action through triggering this response. These findings illuminate a regulatory mechanism for conditional PZA susceptibility and reveal new avenues for enhancing potency of this important drug through targeting activation of the cell envelope stress response. IMPORTANCE For decades, pyrazinamide has served as a cornerstone of tuberculosis therapy. Unlike any other antitubercular drug, pyrazinamide requires an acidic environment to exert its action. Despite its importance, the driver of this conditional susceptibility has remained unknown. In this study, a genome-wide approach revealed that pyrazinamide action is governed by the cell envelope stress response. This observation was validated by orthologous approaches that demonstrate that a central player of this response, SigE, is both necessary and sufficient for potentiation of pyrazinamide action. Moreover, constitutive activation of this response through deletion of the anti-sigma factor gene rseA or exposure of bacilli to drugs that target the cell wall was found to potently drive pyrazinamide susceptibility independent of environmental pH. These findings force a paradigm shift in our understanding of pyrazinamide action and open new avenues for improving diagnostic and therapeutic tools for tuberculosis.
Keywords: cell envelope; drug discovery; drug resistance mechanisms; genomics; metabolism; tuberculosis.
Conflict of interest statement
The authors declare no conflict of interest.
We declare no competing interests of a financial or any other nature.
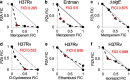
Figures




References
-
- World Health Organization. 2019. Global tuberculosis report 2019.
-
- East African/British Medical Research Councils. 1974. Controlled clinical trial of four short-course (6-month) regimens of pulmonary tuberculosis: third report. Lancet 304:237–240. - PubMed
-
- Maslov DA, Shur KV, Bekker OB, Zakharevich NV, Zaichikova MV, Klimina KM, Smirnova TG, Zhang Y, Chernousova LN, Danilenko VN. 2015. Draft genome sequences of two pyrazinamide-resistant clinical isolates, Mycobacterium tuberculosis 13–4152 and 13–2459. Genome Announc 3:e00758-15. doi: 10.1128/genomeA.00758-15. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

