Bromhexine Hydrochloride Prophylaxis of COVID-19 for Medical Personnel: A Randomized Open-Label Study
- PMID: 35103059
- PMCID: PMC8799951
- DOI: 10.1155/2022/4693121
Bromhexine Hydrochloride Prophylaxis of COVID-19 for Medical Personnel: A Randomized Open-Label Study
Abstract
Background: Bromhexine hydrochloride has been suggested as a TMPRSS2 protease blocker that precludes the penetration of SARS-CoV-2 into cells. We aimed to assess the preventive potential of regular bromhexine hydrochloride intake for COVID-19 risk reduction in medical staff actively involved in the evaluation and treatment of patients with confirmed or suspected SARS-CoV-2 infection.
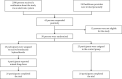
Methods: In a single-centre randomized open-label study, medical staff managing patients with suspected and confirmed COVID-19 were enrolled and followed up for 8 weeks. The study began at the initiation of COVID-19 management in the clinic. The study was prematurely terminated after the enrollment of 50 participants without a history of SARS-CoV-2 infection: 25 were assigned to bromhexine hydrochloride treatment (8 mg 3 times per day), and 25 were controls. The composite primary endpoint was a positive nasopharyngeal swab polymerase chain reaction (PCR) test for SARS-CoV-2 or signs of clinical infection within 28 days and at week 8. Secondary endpoints included time from the first contact with a person with COVID-19 to the appearance of respiratory infection symptoms; the number of days before a first positive SARS-CoV-2 test; the number of asymptomatic participants with a positive nasopharyngeal swab test; the number of symptomatic COVID-19 cases; and adverse events.
Results: The rate of the combined primary endpoint did not differ significantly between the active treatment group (2/25 [8%]) and control group (7/25 [28%]); P=0.07. A fewer number of participants developed symptomatic COVID-19 in the treatment group compared to controls (0/25 vs. 5/25; P=0.02).
Conclusion: Although the study was underpowered, it showed that Bromhexine hydrochloride prophylaxis was associated with a reduced rate of symptomatic COVID-19. The prophylactic treatment was not associated with a lower combined primary endpoint rate, a positive swab PCR test, or COVID-19 (ClinicalTrials.gov number, NCT04405999).
Copyright © 2022 Evgeny N. Mikhaylov et al.
Conflict of interest statement
The authors declare no conflicts of interest in the scope of the present work.
References
Associated data
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous