Reverse re-modelling chronic heart failure by reinstating heart rate variability
- PMID: 35103864
- PMCID: PMC8807455
- DOI: 10.1007/s00395-022-00911-0
Reverse re-modelling chronic heart failure by reinstating heart rate variability
Abstract
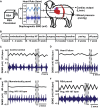
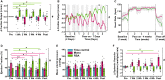
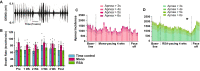
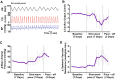
Heart rate variability (HRV) is a crucial indicator of cardiovascular health. Low HRV is correlated with disease severity and mortality in heart failure. Heart rate increases and decreases with each breath in normal physiology termed respiratory sinus arrhythmia (RSA). RSA is highly evolutionarily conserved, most prominent in the young and athletic and is lost in cardiovascular disease. Despite this, current pacemakers either pace the heart in a metronomic fashion or sense activity in the sinus node. If RSA has been lost in cardiovascular disease current pacemakers cannot restore it. We hypothesized that restoration of RSA in heart failure would improve cardiac function. Restoration of RSA in heart failure was assessed in an ovine model of heart failure with reduced ejection fraction. Conscious 24 h recordings were made from three groups, RSA paced (n = 6), monotonically paced (n = 6) and heart failure time control (n = 5). Real-time blood pressure, cardiac output, heart rate and diaphragmatic EMG were recorded in all animals. Respiratory modulated pacing was generated by a proprietary device (Ceryx Medical) to pace the heart with real-time respiratory modulation. RSA pacing substantially increased cardiac output by 1.4 L/min (20%) compared to contemporary (monotonic) pacing. This increase in cardiac output led to a significant decrease in apnoeas associated with heart failure, reversed cardiomyocyte hypertrophy, and restored the T-tubule structure that is essential for force generation. Re-instating RSA in heart failure improves cardiac function through mechanisms of reverse re-modelling; the improvement observed is far greater than that seen with current contemporary therapies. These findings support the concept of re-instating RSA as a regime for patients who require a pacemaker.
Keywords: Cardiac output; Heart failure; Pacemaker; Respiratory sinus arrhythmia.
© 2022. The Author(s).
Conflict of interest statement
Professors Julian Paton and Alain Nogaret are Directors of Ceryx Medical Limited. Dr. Rohit Ramchandra and his lab acknowledge funding from Ceryx Medical Ltd.
Figures


References
-
- Abraham WT, Fisher WG, Smith AL, Delurgio DB, Leon AR, Loh E, Kocovic DZ, Packer M, Clavell AL, Hayes DL, Ellestad M, Trupp RJ, Underwood J, Pickering F, Truex C, McAtee P, Messenger J, Evaluation MSGMIRC Cardiac resynchronization in chronic heart failure. N Engl J Med. 2002;346:1845–1853. doi: 10.1056/NEJMoa013168. - DOI - PubMed
-
- Abraham WT, Kuck KH, Goldsmith RL, Lindenfeld J, Reddy VY, Carson PE, Mann DL, Saville B, Parise H, Chan R, Wiegn P, Hastings JL, Kaplan AJ, Edelmann F, Luthje L, Kahwash R, Tomassoni GF, Gutterman DD, Stagg A, Burkhoff D, Hasenfuss G. A randomized controlled trial to evaluate the safety and efficacy of cardiac contractility modulation. JACC Heart Fail. 2018;6:874–883. doi: 10.1016/j.jchf.2018.04.010. - DOI - PubMed
-
- Abraham WT, Zile MR, Weaver FA, Butter C, Ducharme A, Halbach M, Klug D, Lovett EG, Muller-Ehmsen J, Schafer JE, Senni M, Swarup V, Wachter R, Little WC. Baroreflex activation therapy for the treatment of heart failure with a reduced ejection fraction. JACC Heart Fail. 2015;3:487–496. doi: 10.1016/j.jchf.2015.02.006. - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

