Inhibition of interferon-gamma-stimulated melanoma progression by targeting neuronal nitric oxide synthase (nNOS)
- PMID: 35105915
- PMCID: PMC8807785
- DOI: 10.1038/s41598-022-05394-6
Inhibition of interferon-gamma-stimulated melanoma progression by targeting neuronal nitric oxide synthase (nNOS)
Abstract
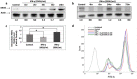
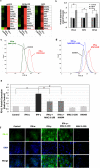
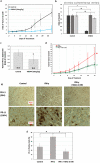
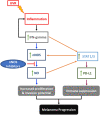
Interferon-gamma (IFN-γ) is shown to stimulate melanoma development and progression. However, the underlying mechanism has not been completely defined. Our study aimed to determine the role of neuronal nitric oxide synthase (nNOS)-mediated signaling in IFN-γ-stimulated melanoma progression and the anti-melanoma effects of novel nNOS inhibitors. Our study shows that IFN-γ markedly induced the expression levels of nNOS in melanoma cells associated with increased intracellular nitric oxide (NO) levels. Co-treatment with novel nNOS inhibitors effectively alleviated IFN-γ-activated STAT1/3. Further, reverse phase protein array (RPPA) analysis demonstrated that IFN-γ induced the expression of HIF1α, c-Myc, and programmed death-ligand 1 (PD-L1), in contrast to IFN-α. Blocking the nNOS-mediated signaling pathway using nNOS-selective inhibitors was shown to effectively diminish IFN-γ-induced PD-L1 expression in melanoma cells. Using a human melanoma xenograft mouse model, the in vivo studies revealed that IFN-γ increased tumor growth compared to control, which was inhibited by the co-administration of nNOS inhibitor MAC-3-190. Another nNOS inhibitor, HH044, was shown to effectively inhibit in vivo tumor growth and was associated with reduced PD-L1 expression levels in melanoma xenografts. Our study demonstrates the important role of nNOS-mediated NO signaling in IFN-γ-stimulated melanoma progression. Targeting nNOS using highly selective small molecular inhibitors is a unique and effective strategy to improve melanoma treatment.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

