Dipeptidase-1 governs renal inflammation during ischemia reperfusion injury
- PMID: 35108057
- PMCID: PMC8809686
- DOI: 10.1126/sciadv.abm0142
Dipeptidase-1 governs renal inflammation during ischemia reperfusion injury
Abstract
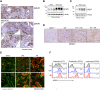
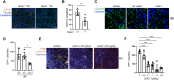
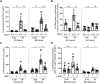
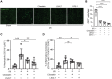
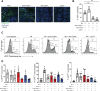
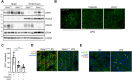
The mechanisms that drive leukocyte recruitment to the kidney are incompletely understood. Dipeptidase-1 (DPEP1) is a major neutrophil adhesion receptor highly expressed on proximal tubular cells and peritubular capillaries of the kidney. Renal ischemia reperfusion injury (IRI) induces robust neutrophil and monocyte recruitment and causes acute kidney injury (AKI). Renal inflammation and the AKI phenotype were attenuated in Dpep1-/- mice or mice pretreated with DPEP1 antagonists, including the LSALT peptide, a nonenzymatic DPEP1 inhibitor. DPEP1 deficiency or inhibition primarily blocked neutrophil adhesion to peritubular capillaries and reduced inflammatory monocyte recruitment to the kidney after IRI. CD44 but not ICAM-1 blockade also decreased neutrophil recruitment to the kidney during IRI and was additive to DPEP1 effects. DPEP1, CD44, and ICAM-1 all contributed to the recruitment of monocyte/macrophages to the kidney following IRI. These results identify DPEP1 as a major leukocyte adhesion receptor in the kidney and potential therapeutic target for AKI.
Figures

References
-
- James M. T., Ghali W. A., Knudtson M. L., Ravani P., Tonelli M., Faris P., Pannu N., Manns B. J., Klarenbach S. W., Hemmelgarn B. R.; Alberta Provincial Project for Outcome Assessment in Coronary Heart Disease (APPROACH) Investigators , Associations between acute kidney injury and cardiovascular and renal outcomes after coronary angiography. Circulation 123, 409–416 (2011). - PubMed
-
- James M. T., Levey A. S., Tonelli M., Tan Z., Barry R., Pannu N., Ravani P., Klarenbach S. W., Manns B. J., Hemmelgarn B. R., Incidence and prognosis of acute kidney diseases and disorders using an integrated approach to laboratory measurements in a universal health care system. JAMA Netw. Open 2, e191795 (2019). - PMC - PubMed
-
- Rewa O., Bagshaw S. M., Acute kidney injury-epidemiology, outcomes and economics. Nat. Rev. Nephrol. 10, 193–207 (2014). - PubMed
-
- Wang Y., Bellomo R., Cardiac surgery-associated acute kidney injury: Risk factors, pathophysiology and treatment. Nat. Rev. Nephrol. 13, 697–711 (2017). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

