Thrombin cleavage of osteopontin initiates osteopontin's tumor-promoting activity
- PMID: 35108449
- PMCID: PMC9289821
- DOI: 10.1111/jth.15663
Thrombin cleavage of osteopontin initiates osteopontin's tumor-promoting activity
Abstract
Background: Osteopontin (OPN) is a multifunctional proinflammatory matricellular protein overexpressed in multiple human cancers and associated with tumor progression and metastases. Thrombin cleavage of OPN reveals a cryptic binding site for α4 β1 and α9 β1 integrins.
Methods: Thrombin cleavage-resistant OPNR153A knock-in (OPN-KI) mice were generated and compared to OPN deficient mice (OPN-KO) and wild type (WT) mice in their ability to support growth of melanoma cells. Flow cytometry was used to analyze tumor infiltrating leukocytes.
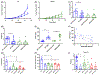
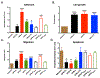
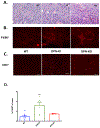
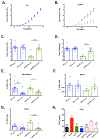
Results: OPN-KI mice engineered with a thrombin cleavage-resistant OPN had reduced B16 melanoma growth and fewer pulmonary metastases than WT mice. The tumor suppression phenotype of the OPN-KI mouse was identical to that observed in OPN-KO mice and was replicated in WT mice by pharmacologic inhibition of thrombin with dabigatran. Tumors isolated from OPN-KI mice had increased tumor-associated macrophages with an altered activation phenotype. Immunodeficient OPN-KI mice (NOG-OPN-KI) or macrophage-depleted OPN-KI mice did not exhibit the tumor suppression phenotype. As B16 cells do not express OPN, thrombin-cleaved fragments of host OPN suppress host antitumor immune response by functionally modulating the tumor-associated macrophages. YUMM3.1 cells, which express OPN, showed less tumor suppression in the OPN-KI and OPN-KO mice than B16 cells, but its growth was suppressed by dabigatran similar to B16 cells.
Conclusions: Thrombin cleavage of OPN, derived from the host and the tumor, initiates OPN's tumor-promoting activity in vivo.
Keywords: B16 melanoma; metastasis; osteopontin; thrombin inhibitor; tumor-associated macrophages.
© 2022 International Society on Thrombosis and Haemostasis.
Conflict of interest statement
Figures



References
-
- Chiodoni C, Colombo MP, Sangaletti S. Matricellular proteins: from homeostasis to inflammation, cancer, and metastasis. Cancer Metastasis Rev. 2010; 29: 295–307. - PubMed
-
- Singhal H, Bautista DS, Tonkin KS, O’Malley FP, Tuck AB, Chambers AF, Harris JF. Elevated plasma osteopontin in metastatic breast cancer associated with increased tumor burden and decreased survival. Clinical cancer research : an official journal of the American Association for Cancer Research. 1997; 3: 605–11. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

