In vitro regeneration and Agrobacterium-mediated genetic transformation of Dragon's Head plant (Lallemantia iberica)
- PMID: 35110618
- PMCID: PMC8810885
- DOI: 10.1038/s41598-022-05776-w
In vitro regeneration and Agrobacterium-mediated genetic transformation of Dragon's Head plant (Lallemantia iberica)
Abstract
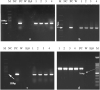
Dragon's head plant (Lallemantia iberica), is a flowering species belongs to the mint family (Lamiaceae). The species contains valuable essential oils, mucilage and oil which are used in pharmaceutical and food industries. Tissue culture is a feasible strategy to attain large-scale production of plantlets with a huge potential to produce plants with superior quality. The objective of this study was to develop a simple and efficient method for regeneration and transformation of L. iberica. To reach this goal, the regeneration ability of various explants including leaf, cotyledonary node, hypocotyl and cotyledon segments was investigated in MS medium supplemented with diverse concentrations of NAA (Naphthalene acetic acid) and BAP (6-Benzyl Amino Purine). According to the results, cotyledonary nodes showed the best regeneration response. The maximum rate of regeneration (and number of induced shoots was achieved in 1 mg l-1 BAP in combination with 0.05 mg l-1 NAA from the cotyledonary nodes. Additionally, through the optimized regeneration technique Agrobacterium-mediated transformation of L. iberica was successfully accomplished. Gene transfer was assessed on leaf samples from regenerated plantlets under a fluorescent microscope to detect the GFP signals. Moreover, transgene integration and its expression were confirmed by PCR and RT-PCR analysis, respectively. The establishment of these efficient regeneration and genetic transformation methods paved the way for further application such as plant improvement, functional analysis and gene editing.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures







Similar articles
-
High frequency regeneration of plants via callus-mediated organogenesis from cotyledon and hypocotyl cultures in a multipurpose tropical tree (Neolamarkia Cadamba).Sci Rep. 2020 Mar 12;10(1):4558. doi: 10.1038/s41598-020-61612-z. Sci Rep. 2020. PMID: 32165694 Free PMC article.
-
Regeneration and transformation of Crambe abyssinica.BMC Plant Biol. 2014 Sep 3;14:235. doi: 10.1186/s12870-014-0235-1. BMC Plant Biol. 2014. PMID: 25195944 Free PMC article.
-
Efficient micropropagation of highly economic, medicinal and ornamental plant Lallemantia iberica (Bieb.) Fisch. and C. A. Mey.Biomed Res Int. 2014;2014:476346. doi: 10.1155/2014/476346. Epub 2014 Aug 27. Biomed Res Int. 2014. PMID: 25247175 Free PMC article.
-
Soybean (Glycine max) transformation using immature cotyledon explants.Methods Mol Biol. 2006;343:397-405. doi: 10.1385/1-59745-130-4:397. Methods Mol Biol. 2006. PMID: 16988362 Review.
-
Advancements in plant transformation: from traditional methods to cutting-edge techniques and emerging model species.Plant Cell Rep. 2024 Oct 29;43(11):273. doi: 10.1007/s00299-024-03359-9. Plant Cell Rep. 2024. PMID: 39467894 Review.
Cited by
-
In vitro regeneration and its histological characteristics of Dioscorea nipponica Makino.Sci Rep. 2022 Nov 1;12(1):18436. doi: 10.1038/s41598-022-22986-4. Sci Rep. 2022. PMID: 36319819 Free PMC article.
-
Protocol for Generating Arabidopsis thaliana Cell Suspension Cultures Under Different Light Conditions.Methods Mol Biol. 2024;2827:145-153. doi: 10.1007/978-1-0716-3954-2_9. Methods Mol Biol. 2024. PMID: 38985267
-
Establishment of Agrobacterium-Mediated Transient Transformation System in Sunflower.Plants (Basel). 2025 Aug 4;14(15):2412. doi: 10.3390/plants14152412. Plants (Basel). 2025. PMID: 40805760 Free PMC article.
-
Efficient callus induction and regeneration of tea plant (Camellia sinensis).Sci Rep. 2025 Jul 23;15(1):26848. doi: 10.1038/s41598-025-12271-5. Sci Rep. 2025. PMID: 40702144 Free PMC article.
-
Enhancing Micropropagation of Adenophora liliifolia: Insights from PGRs, Natural Extracts, and pH Optimization.Plants (Basel). 2024 Jun 23;13(13):1735. doi: 10.3390/plants13131735. Plants (Basel). 2024. PMID: 38999573 Free PMC article.
References
-
- Ursu B, Borcean I. Researches concerning the sowing technology at Lallemantia iberica. Res. J. Agric. Sci. 2012;44(1):168–171.
-
- Overeem A, Buisman G, Derksen J, Cuperus F, Molhoek L, Grisnich W, Goemans C. Seed oils rich in linolenic acid as renewable feedstock for environment-friendly crosslinkers in powder coatings. Ind. Crops Prod. 1999;10:157–165.
-
- Morteza-Semnani K. essential oil composition of Lallemantia iberica Fisch. et CA Mey. J. Essent. Oil Res. 2006;18:164–165.
-
- Amanzadeh Y, Khosravi Dehaghi N, Gohari A, Monsef-Esfahani H, Sadat Ebrahimi S. Antioxidant activity of essential oil of Lallemantia iberica in flowering stage and post-flowering stage. Res. J. Biol. Sci. 2011;6:114–117.
-
- Gunstone, F. & Harwood, J. Occurrence and characterisation of oils and fats, The lipid handbook with CD-ROM. CRC press, pp. 51–156 (2007).
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

