Generalizability of Results from Randomized Controlled Trials in Post-Stroke Physiotherapy
- PMID: 35110812
- PMCID: PMC8781507
- DOI: 10.3138/ptc-2018-0117
Generalizability of Results from Randomized Controlled Trials in Post-Stroke Physiotherapy
Abstract
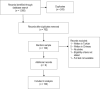
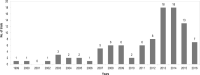
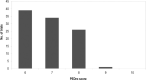
Purpose: The randomized controlled trial (RCT) is considered a reliable experimental design, able to detect the effect of an intervention. However, a criticism frequently levelled at RCTs by clinicians is their lack of generalizability. This study aimed to evaluate the generalizability of findings from RCTs of physiotherapy interventions for individuals with stroke. Method: A sample of RCTs of physiotherapy interventions after stroke indexed in the PEDro database was selected, and the reported inclusion and exclusion criteria were analyzed. Results: We reviewed 100 articles, which included 7,366 participants (41.6% women, with a mean weighted age of 65.5 years). The most frequent criteria for exclusion were comorbidity (83%), cognitive impairments (69%), communication skills (55%), recurrent stroke (53%), low functional level (47%) and being elderly (25%). Conclusions: A variety of cohorts of individuals who have had a stroke are excluded from RCTs published in the field of physiotherapy. Because they represent a substantial proportion of the real-world population with stroke, and consequently treated in clinical practice, more vulnerable cohorts of participants should be included in RCTs.
Objectif : l’essai aléatoire et contrôlé (EAC) est considéré comme une méthodologie expérimentale fiable, en mesure de déterminer l’effet d’une intervention. Cependant, les cliniciens en critiquent souvent l’absence de généralisabilité. La présente étude visait à évaluer la généralisabilité des résultats des EAC sur des interventions de recherche en physiothérapie auprès de personnes ayant subi un accident vasculaire cérébral (AVC). Méthodologie : les auteurs ont sélectionné un échantillon d’EAC sur des interventions de physiothérapie après un AVC indexées dans la base de données PEDro. Ils ont analysé les critères d’inclusion et d’exclusion déclarés. Résultats : les auteurs ont examiné 100 articles, qui incluaient 7 366 participants (41,6 % de femmes, d’un âge moyen pondéré de 65,5 ans). Les critères d’exclusion les plus fréquents étaient la comorbidité (83 %), les déficiences cognitives (69 %), les aptitudes à la communication (55 %), les AVC récurrents (53 %), un faible niveau fonctionnel (47 %) et le fait d’être âgé (25 %). Conclusion : diverses cohortes de personnes qui ont subi un AVC sont exclues des EAC publiés en physiothérapie. Puisqu’elles représentent une forte proportion de la population de personnes qui subissent un AVC en situation réelle et qui sont ensuite traitées en pratique clinique, il faudrait inclure plus de cohortes de participants vulnérables dans les EAC.
Keywords: accident vasculaire cérébral; rehabilitation; reproducibility of results; reproductibilité des résultats; réadaptation; stroke.
© Canadian Physiotherapy Association.
Figures
Similar articles
-
Comparing stroke rehabilitation inpatients and clinical trials eligibility criteria: A secondary chart review analysis revealing that most patients could have been excluded from rehabilitation trials based on comorbidity status.J Multimorb Comorb. 2023 Nov 1;13:26335565231211668. doi: 10.1177/26335565231211668. eCollection 2023 Jan-Dec. J Multimorb Comorb. 2023. PMID: 37927973 Free PMC article.
-
Airway clearance devices for cystic fibrosis: an evidence-based analysis.Ont Health Technol Assess Ser. 2009;9(26):1-50. Epub 2009 Nov 1. Ont Health Technol Assess Ser. 2009. PMID: 23074531 Free PMC article.
-
Methodological Quality of Motor Intervention Randomized Controlled Trials in Stroke Rehabilitation.J Stroke Cerebrovasc Dis. 2016 Feb;25(2):248-53. doi: 10.1016/j.jstrokecerebrovasdis.2015.09.024. Epub 2015 Oct 20. J Stroke Cerebrovasc Dis. 2016. PMID: 26500172
-
Eligibility criteria of randomized controlled trials published in high-impact general medical journals: a systematic sampling review.JAMA. 2007 Mar 21;297(11):1233-40. doi: 10.1001/jama.297.11.1233. JAMA. 2007. PMID: 17374817 Review.
-
Are plain-language summaries included in published reports of evidence about physiotherapy interventions? Analysis of 4421 randomised trials, systematic reviews and guidelines on the Physiotherapy Evidence Database (PEDro).Physiotherapy. 2019 Sep;105(3):354-361. doi: 10.1016/j.physio.2018.11.003. Epub 2018 Nov 15. Physiotherapy. 2019. PMID: 30876718 Review.
Cited by
-
Semi-automated tortuosity measurements confirm generalizability of IMPERATIVE trial results to real-world patients with acute ischemic stroke undergoing thrombectomy.Interv Neuroradiol. 2025 Jul 21:15910199251359089. doi: 10.1177/15910199251359089. Online ahead of print. Interv Neuroradiol. 2025. PMID: 40686305 Free PMC article.
-
Inclusion of adults with conditions that have the potential to affect capacity and or communication in research: triangulation from a mixed-methods study of current practice and values across multiple stakeholders.BMJ Open. 2023 Apr 28;13(4):e068366. doi: 10.1136/bmjopen-2022-068366. BMJ Open. 2023. PMID: 37116998 Free PMC article.
-
Co-design for stroke intervention development: Results of a scoping review.PLoS One. 2024 Feb 14;19(2):e0297162. doi: 10.1371/journal.pone.0297162. eCollection 2024. PLoS One. 2024. PMID: 38354160 Free PMC article.
-
The influence of bias in randomized controlled trials on rehabilitation intervention effect estimates: what we have learned from meta-epidemiological studies.Eur J Phys Rehabil Med. 2024 Feb;60(1):135-144. doi: 10.23736/S1973-9087.23.08310-7. Epub 2023 Dec 12. Eur J Phys Rehabil Med. 2024. PMID: 38088137 Free PMC article.
-
Comparing stroke rehabilitation inpatients and clinical trials eligibility criteria: A secondary chart review analysis revealing that most patients could have been excluded from rehabilitation trials based on comorbidity status.J Multimorb Comorb. 2023 Nov 1;13:26335565231211668. doi: 10.1177/26335565231211668. eCollection 2023 Jan-Dec. J Multimorb Comorb. 2023. PMID: 37927973 Free PMC article.
References
Studies Referenced in Tables 1 and 2
LinkOut - more resources
Full Text Sources