Nε-Lysine Acetylation of the Histone-Like Protein HBsu Regulates the Process of Sporulation and Affects the Resistance Properties of Bacillus subtilis Spores
- PMID: 35111139
- PMCID: PMC8801598
- DOI: 10.3389/fmicb.2021.782815
Nε-Lysine Acetylation of the Histone-Like Protein HBsu Regulates the Process of Sporulation and Affects the Resistance Properties of Bacillus subtilis Spores
Abstract
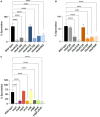
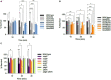
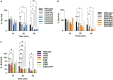
Bacillus subtilis produces dormant, highly resistant endospores in response to extreme environmental stresses or starvation. These spores are capable of persisting in harsh environments for many years, even decades, without essential nutrients. Part of the reason that these spores can survive such extreme conditions is because their chromosomal DNA is well protected from environmental insults. The α/β-type small acid-soluble proteins (SASPs) coat the spore chromosome, which leads to condensation and protection from such insults. The histone-like protein HBsu has been implicated in the packaging of the spore chromosome and is believed to be important in modulating SASP-mediated alterations to the DNA, including supercoiling and stiffness. Previously, we demonstrated that HBsu is acetylated at seven lysine residues, and one physiological function of acetylation is to regulate chromosomal compaction. Here, we investigate if the process of sporulation or the resistance properties of mature spores are influenced by the acetylation state of HBsu. Using our collection of point mutations that mimic the acetylated and unacetylated forms of HBsu, we first determined if acetylation affects the process of sporulation, by determining the overall sporulation frequencies. We found that specific mutations led to decreases in sporulation frequency, suggesting that acetylation of HBsu at some sites, but not all, is required to regulate the process of sporulation. Next, we determined if the spores produced from the mutant strains were more susceptible to heat, ultraviolet (UV) radiation and formaldehyde exposure. We again found that altering acetylation at specific sites led to less resistance to these stresses, suggesting that proper HBsu acetylation is important for chromosomal packaging and protection in the mature spore. Interestingly, the specific acetylation patterns were different for the sporulation process and resistance properties of spores, which is consistent with the notion that a histone-like code exists in bacteria. We propose that specific acetylation patterns of HBsu are required to ensure proper chromosomal arrangement, packaging, and protection during the process of sporulation.
Keywords: KAT; KDAC; SASP; acetyl; acetylation; bacteria; endospore; post-translational modification (PTM).
Copyright © 2022 Luu, Mott, Schreiber, Giovinco, Betchen and Carabetta.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Nε-lysine acetylation of the histone-like protein HBsu influences antibiotic survival and persistence in Bacillus subtilis.Front Microbiol. 2024 May 21;15:1356733. doi: 10.3389/fmicb.2024.1356733. eCollection 2024. Front Microbiol. 2024. PMID: 38835483 Free PMC article.
-
YfmK is an Nε-lysine acetyltransferase that directly acetylates the histone-like protein HBsu in Bacillus subtilis.Proc Natl Acad Sci U S A. 2019 Feb 26;116(9):3752-3757. doi: 10.1073/pnas.1815511116. Epub 2019 Feb 11. Proc Natl Acad Sci U S A. 2019. PMID: 30808761 Free PMC article.
-
The Bacillus subtilis HBsu protein modifies the effects of alpha/beta-type, small acid-soluble spore proteins on DNA.J Bacteriol. 2000 Apr;182(7):1942-8. doi: 10.1128/JB.182.7.1942-1948.2000. J Bacteriol. 2000. PMID: 10715001 Free PMC article.
-
Spores of Bacillus subtilis: their resistance to and killing by radiation, heat and chemicals.J Appl Microbiol. 2006 Sep;101(3):514-25. doi: 10.1111/j.1365-2672.2005.02736.x. J Appl Microbiol. 2006. PMID: 16907802 Review.
-
Addressing the Possibility of a Histone-Like Code in Bacteria.J Proteome Res. 2021 Jan 1;20(1):27-37. doi: 10.1021/acs.jproteome.0c00442. Epub 2020 Oct 2. J Proteome Res. 2021. PMID: 32962352 Free PMC article. Review.
Cited by
-
Nε-lysine acetylation of the histone-like protein HBsu influences antibiotic survival and persistence in Bacillus subtilis.Front Microbiol. 2024 May 21;15:1356733. doi: 10.3389/fmicb.2024.1356733. eCollection 2024. Front Microbiol. 2024. PMID: 38835483 Free PMC article.
-
A Sporulation-Specific sRNA Bvs196 Contributing to the Developing Spore in Bacillus velezensis.Microorganisms. 2022 May 12;10(5):1015. doi: 10.3390/microorganisms10051015. Microorganisms. 2022. PMID: 35630459 Free PMC article.
-
Epigenetic-Mediated Antimicrobial Resistance: Host versus Pathogen Epigenetic Alterations.Antibiotics (Basel). 2022 Jun 16;11(6):809. doi: 10.3390/antibiotics11060809. Antibiotics (Basel). 2022. PMID: 35740215 Free PMC article. Review.
-
Acetylomics reveals an extensive acetylation diversity within Pseudomonas aeruginosa.Microlife. 2024 Sep 14;5:uqae018. doi: 10.1093/femsml/uqae018. eCollection 2024. Microlife. 2024. PMID: 39464744 Free PMC article.
References
-
- Banks J., Morgan S., Stringer M. (1988). Inhibition of heated Bacillus spores by combinations of potassium sorbate, sodium benzoate, pH and organic acids. Lebensmittel-Wissenschaft Technologie 21 250–255.
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

