Internal Stark effect of single-molecule fluorescence
- PMID: 35115513
- PMCID: PMC8813982
- DOI: 10.1038/s41467-022-28241-8
Internal Stark effect of single-molecule fluorescence
Abstract
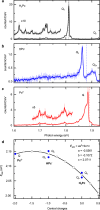
The optical properties of chromophores can be efficiently tuned by electrostatic fields generated in their close environment, a phenomenon that plays a central role for the optimization of complex functions within living organisms where it is known as internal Stark effect (ISE). Here, we realised an ISE experiment at the lowest possible scale, by monitoring the Stark shift generated by charges confined within a single chromophore on its emission energy. To this end, a scanning tunneling microscope (STM) functioning at cryogenic temperatures is used to sequentially remove the two central protons of a free-base phthalocyanine chromophore deposited on a NaCl-covered Ag(111) surface. STM-induced fluorescence measurements reveal spectral shifts that are associated to the electrostatic field generated by the internal charges remaining in the chromophores upon deprotonation.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



References
-
- Honig B, et al. An external point-charge model for wavelength regulation in visual pigments. J. Am. Chem. Soc. 1979;101:7084–7086.
-
- Gottfried D, Steffen M, Boxer S. Large protein-induced dipoles for a symmetric carotenoid in a photosynthetic antenna complex. Science. 1991;251:662–665. - PubMed
-
- Lockhart D, Kim P. Internal Stark effect measurement of the electric field at the amino terminus of an alpha helix. Science. 1992;257:947–951. - PubMed
-
- Boxer SG. Stark realities. J. Phys. Chem. B. 2009;113:2972–2983. - PubMed
Publication types
LinkOut - more resources
Full Text Sources

