Anatomical variability, multi-modal coordinate systems, and precision targeting in the marmoset brain
- PMID: 35122965
- PMCID: PMC8948178
- DOI: 10.1016/j.neuroimage.2022.118965
Anatomical variability, multi-modal coordinate systems, and precision targeting in the marmoset brain
Abstract
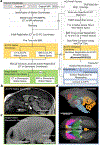
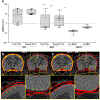
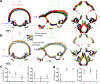
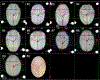
Localising accurate brain regions needs careful evaluation in each experimental species due to their individual variability. However, the function and connectivity of brain areas is commonly studied using a single-subject cranial landmark-based stereotactic atlas in animal neuroscience. Here, we address this issue in a small primate, the common marmoset, which is increasingly widely used in systems neuroscience. We developed a non-invasive multi-modal neuroimaging-based targeting pipeline, which accounts for intersubject anatomical variability in cranial and cortical landmarks in marmosets. This methodology allowed creation of multi-modal templates (MarmosetRIKEN20) including head CT and brain MR images, embedded in coordinate systems of anterior and posterior commissures (AC-PC) and CIFTI grayordinates. We found that the horizontal plane of the stereotactic coordinate was significantly rotated in pitch relative to the AC-PC coordinate system (10 degrees, frontal downwards), and had a significant bias and uncertainty due to positioning procedures. We also found that many common cranial and brain landmarks (e.g., bregma, intraparietal sulcus) vary in location across subjects and are substantial relative to average marmoset cortical area dimensions. Combining the neuroimaging-based targeting pipeline with robot-guided surgery enabled proof-of-concept targeting of deep brain structures with an accuracy of 0.2 mm. Altogether, our findings demonstrate substantial intersubject variability in marmoset brain and cranial landmarks, implying that subject-specific neuroimaging-based localization is needed for precision targeting in marmosets. The population-based templates and atlases in grayordinates, created for the first time in marmoset monkeys, should help bridging between macroscale and microscale analyses.
Keywords: Marmoset; brain; coordinates; cranium; neurosurgery; subject variability.
Copyright © 2022. Published by Elsevier Inc.
Conflict of interest statement
Declaration of Competing Interest Stephen Frey is employed by Rogue Research Inc. All the other authors declare no competing financial interests.
Figures
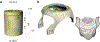
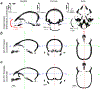





References
-
- Arun KS, Huang TS, Blostein SD, 1987. Least-squares fitting of two 3-d point sets. IEEE Trans. Pattern Anal. Mach. Intell. 9, 698–700 - PubMed
-
- Atsumi I, Kurata M, Sakaki H, 2013. Comparative study on ocular anatomical features among rabbits, beagle dogs and cynomolgus monkeys. Anim. Eye Res. 32, 35–41
-
- Autio JA, Glasser MF, Ose T, Donahue CJ, Bastiani M, Ohno M, Kawabata Y, Urushibata Y, Murata K, Nishigori K, Yamaguchi M, Hori Y, Yoshida A, Go Y, Coalson TS, Jbabdi S, Sotiropoulos SN, Kennedy H, Smith S, Van Essen DC, Hayashi T, 2020. Towards HCP-style Macaque Connectomes: 24-channel 3T multi--array coil, MRI sequences and preprocessing. Neuroimage 116800 - PMC - PubMed
-
- Bowden DM, Martin MF, 2000. Primate Brain Maps: Structure of the Macaque Brain: A Laboratory Guide with Original Brain Sections, Printed Atlas and Electronic Templates for Data and Schematics, 1st ed. Elsevier Science
-
- Burman KJ, Bakola S, Richardson KE, Reser DH, Rosa MGP, 2014. Patterns of afferent input to the caudal and rostral areas of the dorsal premotor cortex (6DC and 6DR) in the marmoset monkey. J. Comp. Neurol. 522, 3683–3716 - PubMed

