1,3,4-Thiadiazole Scaffold: As Anti-Epileptic Agents
- PMID: 35127639
- PMCID: PMC8814426
- DOI: 10.3389/fchem.2021.671212
1,3,4-Thiadiazole Scaffold: As Anti-Epileptic Agents
Abstract
A wide range of biological activities is exhibited by 1,3,4-thiadiazole moiety such as antidiabetic, anticancer, anti-inflammatory, anticonvulsant, antiviral, antihypertensive, and antimicrobial. To date, drugs such as butazolamide, and acetazolamide. Several modifications have been done in the 1,3,4-thiadiazole moiety which showed good potency as anticonvulsant agents which are highly effective and have less toxicity. After in-depth literature survey in this review, we have compiled various derivatives of 1,3,4-thiadiazole scaffold as anticonvulsant agents.
Keywords: 1,3,4 thiadiazole; anti-convulsant; anti-hypertensive; pharmacophoric pattern; structural activity relationship.
Copyright © 2022 Anthwal and Nain.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



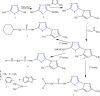
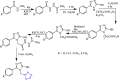

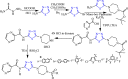
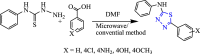
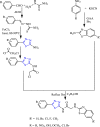


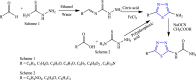
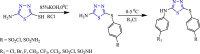
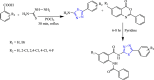
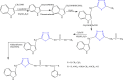
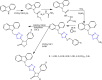

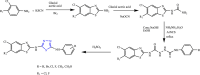


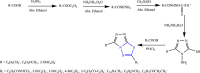
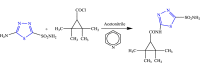
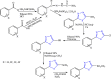
References
-
- Aliyu A., Idris A. Y., Musa A. M., Hamza A. N., Ahmadu J. O., Shehu A., et al. (2021). Design, Synthesis, and In-Vivo Anticonvulsant Evaluation of 5-[(E)-(3, 4, 5-trimethoxybenzylidene) Amino]-1, 3, 4-Thiadiazole-2-Thiol. IJSGS 7 (20), 120–127.
-
- Almasirad A., Vousooghi N., Tabatabai S. A., Kebriaeezadeh A., Shafiee A. (2007). Synthesis, Anticonvulsant, and Muscle Relaxant Activities of Substituted 1, 3, 4-oxadiazole, 1, 3, 4-thiadiazole, and 1, 2, 4-triazole. Acta Chim. Slow 54 (2), 317–324.
-
- Belanger C., P. (1979). 2-(4-substituted-1,2,5-thiadiazole-3-yloxy)-acetaldehydes. U.S. Patent No 4,145,550. Washington, DC: U.S. Patent and Trademark Office.
-
- Bhandari S. V., Deshmane B. J., Dangare S. C., Gore S. T., Raparti V. T., Khachane C. V., et al. (2008). Anticonvulsant Activities of Some Novel 3-[5-substituted, 3, 4-Thiadiazole-Yl]-2-Styryl Quinazoline-4 (3h)-Ones. Pharmacologyonline 2, 604–613.
-
- Bhattacharya S., Kashaw V. (2019). Design, Synthesis, and Anticonvulsant Potential of (E)-3-(5-(substituted Aminomethyl)-1, 3, 4-Thiadiazol-2-Yl)-2-Substituted Styrylquinazolin-4-(3h)-One. J. Drug Deliv. Ther. 9 (2), 591–602.
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

