Optimized HPLC method to elucidate the complex purinergic signaling dynamics that regulate ATP, ADP, AMP, and adenosine levels in human blood
- PMID: 35132577
- PMCID: PMC9123122
- DOI: 10.1007/s11302-022-09842-w
Optimized HPLC method to elucidate the complex purinergic signaling dynamics that regulate ATP, ADP, AMP, and adenosine levels in human blood
Abstract
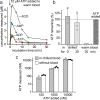
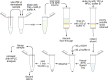
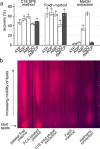
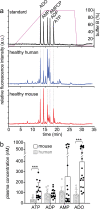
ATP released into the bloodstream regulates immune responses and other physiological functions. Excessive accumulation of extracellular ATP interferes with these functions, and elevated plasma ATP levels could indicate infections and other pathological disorders. However, there is considerable disagreement about what constitutes normal plasma ATP levels. Therefore, we optimized a method to accurately assess ATP concentrations in blood. We found that rapid chilling of heparinized blood samples is essential to preserve in vivo ATP levels and that differential centrifugation minimizes inadvertent ATP release due to cell damage and mechanical stress. Plasma samples were stabilized with perchloric acid, etheno-derivatized, and delipidated for sensitive analysis of ATP and related compounds using high-performance liquid chromatography (HPLC) and fluorescence detection. We measured 33 ± 20 nM ATP, 90 ± 45 nM ADP, 100 ± 55 nM AMP, and 81 ± 51 nM adenosine in the blood of healthy human adults (n = 10). In critically ill patients, ATP levels were 6 times higher than in healthy subjects. The anticoagulant greatly affected results. ATP levels were nearly 8 times higher in EDTA plasma than in heparin plasma, while AMP levels were 3 times lower and adenosine was entirely absent in EDTA plasma. If EDTA blood was not immediately chilled, ATP, ADP, and AMP levels continued to rise, which indicates that EDTA interferes with the endogenous mechanisms that regulate plasma adenylate levels. Our optimized method eliminates artifacts that prevent accurate determination of plasma adenylates and will be useful for studying mechanisms that regulate adenylate levels and for monitoring of pathological processes in patients with infections and other diseases.
Keywords: ATP; Adenosine; HPLC; Plasma adenylates; Purinergic signaling.
© 2022. The Author(s), under exclusive licence to Springer Nature B.V.
Conflict of interest statement
Carola Ledderose declares that she has no conflict of interest.
Eleftheria-Angeliki Valsami declares that she has no conflict of interest.
Wolfgang G. Junger declares that he has no conflict of interest.
Figures




Similar articles
-
Online cleanup of accelerated solvent extractions for determination of adenosine 5'-triphosphate (ATP), adenosine 5'-diphosphate (ADP), and adenosine 5'-monophosphate (AMP) in royal jelly using high-performance liquid chromatography.J Agric Food Chem. 2009 Jun 10;57(11):4500-5. doi: 10.1021/jf900853q. J Agric Food Chem. 2009. PMID: 19435312
-
ATP, ADP and AMP in plasma from peripheral venous blood.Clin Chim Acta. 1984 Nov 15;143(2):91-8. doi: 10.1016/0009-8981(84)90216-x. Clin Chim Acta. 1984. PMID: 6096041
-
Determination of adenosine phosphates in mouse myocardium tissue by HPLC with UV detection and using porous graphite carbon column.J Chromatogr B Analyt Technol Biomed Life Sci. 2020 May 15;1145:122110. doi: 10.1016/j.jchromb.2020.122110. Epub 2020 Apr 11. J Chromatogr B Analyt Technol Biomed Life Sci. 2020. PMID: 32315974
-
[High performance liquid chromatogram (HPLC) determination of adenosine phosphates in rat myocardium].Beijing Da Xue Xue Bao Yi Xue Ban. 2005 Apr 18;37(2):201-2. Beijing Da Xue Xue Bao Yi Xue Ban. 2005. PMID: 15841156 Chinese.
-
Extracellular ATP/adenosine dynamics in the brain and its role in health and disease.Front Cell Dev Biol. 2024 Jan 18;11:1343653. doi: 10.3389/fcell.2023.1343653. eCollection 2023. Front Cell Dev Biol. 2024. PMID: 38304611 Free PMC article. Review.
Cited by
-
Advancements in Adenine Nucleotides Extraction and Quantification from a Single Drop of Human Blood.Molecules. 2024 Nov 28;29(23):5630. doi: 10.3390/molecules29235630. Molecules. 2024. PMID: 39683788 Free PMC article.
-
Simultaneous Recognition and Detection of Adenosine Phosphates by Machine Learning Analysis for Surface-Enhanced Raman Scattering Spectral Data.Sensors (Basel). 2024 Oct 15;24(20):6648. doi: 10.3390/s24206648. Sensors (Basel). 2024. PMID: 39460128 Free PMC article.
-
Disulfiram induces redox imbalance and perturbations in central glucose catabolism and metal homeostasis to inhibit the growth of Staphylococcus aureus.Sci Rep. 2025 May 5;15(1):15658. doi: 10.1038/s41598-025-00078-3. Sci Rep. 2025. PMID: 40325037 Free PMC article.
-
Impaired ATP hydrolysis in blood plasma contributes to age-related neutrophil dysfunction.Immun Ageing. 2024 Jul 3;21(1):45. doi: 10.1186/s12979-024-00441-4. Immun Ageing. 2024. PMID: 38961477 Free PMC article.
-
A liquid chromatography-tandem mass spectrometry based method for the quantification of adenosine nucleotides and NAD precursors and products in various biological samples.Front Immunol. 2023 Sep 20;14:1250762. doi: 10.3389/fimmu.2023.1250762. eCollection 2023. Front Immunol. 2023. PMID: 37799723 Free PMC article.
References
-
- Ralevic V, Burnstock G. Receptors for purines and pyrimidines. Pharmacol Rev. 1998;50:413–492. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

