Unanticipated daytime melatonin secretion on a simulated night shift schedule generates a distinctive 24-h melatonin rhythm with antiphasic daytime and nighttime peaks
- PMID: 35133678
- PMCID: PMC8930611
- DOI: 10.1111/jpi.12791
Unanticipated daytime melatonin secretion on a simulated night shift schedule generates a distinctive 24-h melatonin rhythm with antiphasic daytime and nighttime peaks
Abstract
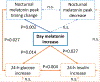
The daily rhythm of plasma melatonin concentrations is typically unimodal, with one broad peak during the circadian night and near-undetectable levels during the circadian day. Light at night acutely suppresses melatonin secretion and phase shifts its endogenous circadian rhythm. In contrast, exposure to darkness during the circadian day has not generally been reported to increase circulating melatonin concentrations acutely. Here, in a highly-controlled simulated night shift protocol with 12-h inverted behavioral/environmental cycles, we unexpectedly found that circulating melatonin levels were significantly increased during daytime sleep (p < .0001). This resulted in a secondary melatonin peak during the circadian day in addition to the primary peak during the circadian night, when sleep occurred during the circadian day following an overnight shift. This distinctive diurnal melatonin rhythm with antiphasic peaks could not be readily anticipated from the behavioral/environmental factors in the protocol (e.g., light exposure, posture, diet, activity) or from current mathematical model simulations of circadian pacemaker output. The observation, therefore, challenges our current understanding of underlying physiological mechanisms that regulate melatonin secretion. Interestingly, the increase in melatonin concentration observed during daytime sleep was positively correlated with the change in timing of melatonin nighttime peak (p = .002), but not with the degree of light-induced melatonin suppression during nighttime wakefulness (p = .92). Both the increase in daytime melatonin concentrations and the change in the timing of the nighttime peak became larger after repeated exposure to simulated night shifts (p = .002 and p = .006, respectively). Furthermore, we found that melatonin secretion during daytime sleep was positively associated with an increase in 24-h glucose and insulin levels during the night shift protocol (p = .014 and p = .027, respectively). Future studies are needed to elucidate the key factor(s) driving the unexpected daytime melatonin secretion and the melatonin rhythm with antiphasic peaks during shifted sleep/wake schedules, the underlying mechanisms of their relationship with glucose metabolism, and the relevance for diabetes risk among shift workers.
Keywords: circadian pacemaker; glucose metabolism; melatonin; night shift.
© 2022 John Wiley & Sons A/S. Published by John Wiley & Sons Ltd.
Conflict of interest statement
Figures

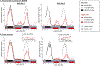

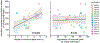

Similar articles
-
A compromise phase position for permanent night shift workers: circadian phase after two night shifts with scheduled sleep and light/dark exposure.Chronobiol Int. 2006;23(4):859-75. doi: 10.1080/07420520600827160. Chronobiol Int. 2006. PMID: 16887753
-
Circadian phase, sleepiness, and light exposure assessment in night workers with and without shift work disorder.Chronobiol Int. 2012 Aug;29(7):928-36. doi: 10.3109/07420528.2012.699356. Chronobiol Int. 2012. PMID: 22823876
-
Progressive decrease of melatonin production over consecutive days of simulated night work.Chronobiol Int. 2014 Dec;31(10):1231-8. doi: 10.3109/07420528.2014.957304. Epub 2014 Sep 15. Chronobiol Int. 2014. PMID: 25222345
-
Disturbance of the Circadian System in Shift Work and Its Health Impact.J Biol Rhythms. 2022 Feb;37(1):3-28. doi: 10.1177/07487304211064218. Epub 2021 Dec 30. J Biol Rhythms. 2022. PMID: 34969316 Free PMC article. Review.
-
Melatonin as a chronobiotic: treatment of circadian desynchrony in night workers and the blind.J Biol Rhythms. 1997 Dec;12(6):595-603. doi: 10.1177/074873049701200615. J Biol Rhythms. 1997. PMID: 9406035 Review.
Cited by
-
Exploring the Causal Association between Morning Diurnal Preference and Psychiatric Disorders: A Bidirectional Two-Sample Mendelian Randomization Analysis.Life (Basel). 2024 Sep 25;14(10):1225. doi: 10.3390/life14101225. Life (Basel). 2024. PMID: 39459525 Free PMC article.
-
The role of melatonin in the development of postmenopausal osteoporosis.Front Pharmacol. 2022 Oct 7;13:975181. doi: 10.3389/fphar.2022.975181. eCollection 2022. Front Pharmacol. 2022. PMID: 36278157 Free PMC article. Review.
-
Melatonin in Glaucoma: Integrative Mechanisms of Intraocular Pressure Control and Neuroprotection.Biomedicines. 2025 May 16;13(5):1213. doi: 10.3390/biomedicines13051213. Biomedicines. 2025. PMID: 40427040 Free PMC article. Review.
-
Actigraphy-Based Sleep Detection: Validation with Polysomnography and Comparison of Performance for Nighttime and Daytime Sleep During Simulated Shift Work.Nat Sci Sleep. 2022 Oct 14;14:1801-1816. doi: 10.2147/NSS.S373107. eCollection 2022. Nat Sci Sleep. 2022. PMID: 36275180 Free PMC article.
References
-
- Kalsbeek A, et al., Melatonin sees the light: blocking GABA-ergic transmission in the paraventricular nucleus induces daytime secretion of melatonin. Eur J Neurosci, 2000. 12(9): p. 3146–54. - PubMed
-
- Perreau-Lenz S, et al., Suprachiasmatic control of melatonin synthesis in rats: inhibitory and stimulatory mechanisms. Eur J Neurosci, 2003. 17(2): p. 221–8. - PubMed
-
- Jewett ME, Kronauer RE, and Czeisler CA, Light-induced suppression of endogenous circadian amplitude in humans. Nature, 1991. 350(6313): p. 59–62. - PubMed
-
- Reppert SM, et al., The effects of environmental lighting on the daily melatonin rhythm in primate cerebrospinal fluid. Brain Res, 1981. 223(2): p. 313–23. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

