A bacterial endosymbiont of the fungus Rhizopus microsporus drives phagocyte evasion and opportunistic virulence
- PMID: 35134329
- PMCID: PMC8926845
- DOI: 10.1016/j.cub.2022.01.028
A bacterial endosymbiont of the fungus Rhizopus microsporus drives phagocyte evasion and opportunistic virulence
Abstract
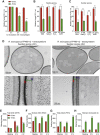
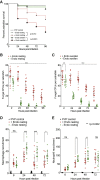
Opportunistic infections by environmental fungi are a growing clinical problem, driven by an increasing population of people with immunocompromising conditions. Spores of the Mucorales order are ubiquitous in the environment but can also cause acute invasive infections in humans through germination and evasion of the mammalian host immune system. How they achieve this and the evolutionary drivers underlying the acquisition of virulence mechanisms are poorly understood. Here, we show that a clinical isolate of Rhizopus microsporus contains a Ralstonia pickettii bacterial endosymbiont required for virulence in both zebrafish and mice and that this endosymbiosis enables the secretion of factors that potently suppress growth of the soil amoeba Dictyostelium discoideum, as well as their ability to engulf and kill other microbes. As amoebas are natural environmental predators of both bacteria and fungi, we propose that this tri-kingdom interaction contributes to establishing endosymbiosis and the acquisition of anti-phagocyte activity. Importantly, we show that this activity also protects fungal spores from phagocytosis and clearance by human macrophages, and endosymbiont removal renders the fungal spores avirulent in vivo. Together, these findings describe a new role for a bacterial endosymbiont in Rhizopus microsporus pathogenesis in animals and suggest a mechanism of virulence acquisition through environmental interactions with amoebas.
Keywords: Dictyostelium; Murcomycete; Ralstonia; Rhizopus; endosymbiosis; evolution; fungal pathogenesis; soil microbiology.
Copyright © 2022 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The authors declare no competing interests.
Figures





Similar articles
-
Exploring Virulence Determinants of Filamentous Fungal Pathogens through Interactions with Soil Amoebae.Front Cell Infect Microbiol. 2017 Dec 5;7:497. doi: 10.3389/fcimb.2017.00497. eCollection 2017. Front Cell Infect Microbiol. 2017. PMID: 29259922 Free PMC article. Review.
-
Virulence determinants of the human pathogenic fungus Aspergillus fumigatus protect against soil amoeba predation.Environ Microbiol. 2015 Aug;17(8):2858-69. doi: 10.1111/1462-2920.12808. Epub 2015 Mar 27. Environ Microbiol. 2015. PMID: 25684622
-
Conidial Melanin of the Human-Pathogenic Fungus Aspergillus fumigatus Disrupts Cell Autonomous Defenses in Amoebae.mBio. 2020 May 26;11(3):e00862-20. doi: 10.1128/mBio.00862-20. mBio. 2020. PMID: 32457245 Free PMC article.
-
Endosymbiosis: the evil within.Curr Biol. 2007 Jun 5;17(11):R408-10. doi: 10.1016/j.cub.2007.04.001. Curr Biol. 2007. PMID: 17550764
-
Pulmonary defense mechanisms against opportunistic fungal pathogens.Immunol Ser. 1989;47:243-71. Immunol Ser. 1989. PMID: 2490078 Review.
Cited by
-
Fungal spore swelling and germination are restricted by the macrophage phagolysosome.Fungal Biol. 2023 Sep;127(9):1291-1297. doi: 10.1016/j.funbio.2023.08.002. Epub 2023 Aug 12. Fungal Biol. 2023. PMID: 37821151 Free PMC article.
-
Genomic evidence of symbiotic adaptations in fungus-associated bacteria.iScience. 2025 Mar 20;28(4):112253. doi: 10.1016/j.isci.2025.112253. eCollection 2025 Apr 18. iScience. 2025. PMID: 40290873 Free PMC article.
-
Endofungal bacteria as hidden facilitators of biotic interactions.ISME J. 2025 Jan 2;19(1):wraf128. doi: 10.1093/ismejo/wraf128. ISME J. 2025. PMID: 40581745 Free PMC article. Review.
-
Microbiota manipulation through the secretion of effector proteins is fundamental to the wealth of lifestyles in the fungal kingdom.FEMS Microbiol Rev. 2022 Sep 2;46(5):fuac022. doi: 10.1093/femsre/fuac022. FEMS Microbiol Rev. 2022. PMID: 35604874 Free PMC article. Review.
-
Symbiotic and toxinogenic Rhizopus spp. isolated from soils of different papaya producing regions in Mexico.Front Fungal Biol. 2022 Oct 24;3:893700. doi: 10.3389/ffunb.2022.893700. eCollection 2022. Front Fungal Biol. 2022. PMID: 37746220 Free PMC article.
References
-
- Chamilos G., Ganguly D., Lande R., Gregorio J., Meller S., Goldman W.E., Gilliet M., Kontoyiannis D.P. Generation of IL-23 producing dendritic cells (DCs) by airborne fungi regulates fungal pathogenicity via the induction of T(H)-17 responses. PLoS One. 2010;5 doi: 10.1371/journal.pone.0012955. - DOI - PMC - PubMed
-
- Petraitis V., Petraitiene R., Antachopoulos C., Hughes J.E., Cotton M.P., Kasai M., Harrington S., Gamaletsou M.N., Bacher J.D., Kontoyiannis D.P., et al. Increased virulence of cunninghamella bertholletiae in experimental pulmonary mucormycosis: correlation with circulating molecular biomarkers, sporangiospore germination and hyphal metabolism. Med. Mycol. 2013;51:72–82. doi: 10.3109/13693786.2012.690107. - DOI - PubMed
Publication types
MeSH terms
Supplementary concepts
Grants and funding
- BB/S010122/1/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- 211241/Z/18/Z/WT_/Wellcome Trust/United Kingdom
- R01 AI063503/AI/NIAID NIH HHS/United States
- BB/M014525/1/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- 102705/Z/13/Z/WT_/Wellcome Trust/United Kingdom
- BB/M01116X/1/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- 102705 /WT_/Wellcome Trust/United Kingdom
- MR/N006364/2/MRC_/Medical Research Council/United Kingdom
- 108387/Z/15/Z/WT_/Wellcome Trust/United Kingdom
- 097377 /WT_/Wellcome Trust/United Kingdom
- MR/V033417/1/MRC_/Medical Research Council/United Kingdom
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

