Satiety Associated with Calorie Restriction and Time-Restricted Feeding: Central Neuroendocrine Integration
- PMID: 35134815
- PMCID: PMC9156369
- DOI: 10.1093/advances/nmac011
Satiety Associated with Calorie Restriction and Time-Restricted Feeding: Central Neuroendocrine Integration
Abstract
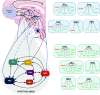
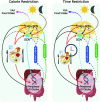
This review focuses on summarizing current knowledge on how time-restricted feeding (TRF) and continuous caloric restriction (CR) affect central neuroendocrine systems involved in regulating satiety. Several interconnected regions of the hypothalamus, brainstem, and cortical areas of the brain are involved in the regulation of satiety. Following CR and TRF, the increase in hunger and reduction in satiety signals of the melanocortin system [neuropeptide Y (NPY), proopiomelanocortin (POMC), and agouti-related peptide (AgRP)] appear similar between CR and TRF protocols, as do the dopaminergic responses in the mesocorticolimbic circuit. However, ghrelin and leptin signaling via the melanocortin system appears to improve energy balance signals and reduce hyperphagia following TRF, which has not been reported in CR. In addition to satiety systems, CR and TRF also influence circadian rhythms. CR influences the suprachiasmatic nucleus (SCN) or the primary circadian clock as seen by increased clock gene expression. In contrast, TRF appears to affect both the SCN and the peripheral clocks, as seen by phasic changes in the non-SCN (potentially the elusive food entrainable oscillator) and metabolic clocks. The peripheral clocks are influenced by the primary circadian clock but are also entrained by food timing, sleep timing, and other lifestyle parameters, which can supersede the metabolic processes that are regulated by the primary circadian clock. Taken together, TRF influences hunger/satiety, energy balance systems, and circadian rhythms, suggesting a role for adherence to CR in the long run if implemented using the TRF approach. However, these suggestions are based on only a few studies, and future investigations that use standardized protocols for the evaluation of the effect of these diet patterns (time, duration, meal composition, sufficiently powered) are necessary to verify these preliminary observations.
Keywords: calorie restriction; circadian rhythms; hypothalamus; light-entrainable oscillator; peripheral oscillators; satiety; time-restricted feeding.
Published by Oxford University Press on behalf of the American Society for Nutrition 2022.
Figures
Similar articles
-
Misalignment of Circadian Rhythms in Diet-Induced Obesity.Adv Exp Med Biol. 2024;1460:27-71. doi: 10.1007/978-3-031-63657-8_2. Adv Exp Med Biol. 2024. PMID: 39287848 Review.
-
Satiety Associated with Calorie Restriction and Time-Restricted Feeding: Peripheral Hormones.Adv Nutr. 2022 Jun 1;13(3):792-820. doi: 10.1093/advances/nmac014. Adv Nutr. 2022. PMID: 35191467 Free PMC article. Review.
-
Natural food intake patterns have little synchronizing effect on peripheral circadian clocks.BMC Biol. 2020 Nov 6;18(1):160. doi: 10.1186/s12915-020-00872-7. BMC Biol. 2020. PMID: 33158435 Free PMC article.
-
Circadian clocks: setting time by food.J Neuroendocrinol. 2007 Feb;19(2):127-37. doi: 10.1111/j.1365-2826.2006.01510.x. J Neuroendocrinol. 2007. PMID: 17214875 Review.
-
Circadian and photic regulation of clock and clock-controlled proteins in the suprachiasmatic nuclei of calorie-restricted mice.Eur J Neurosci. 2007 Jun;25(12):3691-701. doi: 10.1111/j.1460-9568.2007.05626.x. Eur J Neurosci. 2007. PMID: 17610588
Cited by
-
Time-restricted eating improves health because of energy deficit and circadian rhythm: A systematic review and meta-analysis.iScience. 2024 Jan 26;27(2):109000. doi: 10.1016/j.isci.2024.109000. eCollection 2024 Feb 16. iScience. 2024. PMID: 38357669 Free PMC article.
-
Time Restricted Eating: A Valuable Alternative to Calorie Restriction for Addressing Obesity?Curr Obes Rep. 2025 Feb 3;14(1):17. doi: 10.1007/s13679-025-00609-z. Curr Obes Rep. 2025. PMID: 39899119 Free PMC article. Review.
-
Fasting diets: what are the impacts on eating behaviors, sleep, mood, and well-being?Front Nutr. 2024 Jan 9;10:1256101. doi: 10.3389/fnut.2023.1256101. eCollection 2023. Front Nutr. 2024. PMID: 38264193 Free PMC article. Review.
-
Circadian disruption of feeding-fasting rhythm and its consequences for metabolic, immune, cancer, and cognitive processes.Biomed J. 2025 Jun;48(3):100827. doi: 10.1016/j.bj.2025.100827. Epub 2025 Jan 3. Biomed J. 2025. PMID: 39756653 Free PMC article. Review.
-
Circadian Interventions in Preclinical Models of Huntington's Disease: A Narrative Review.Biomedicines. 2024 Aug 6;12(8):1777. doi: 10.3390/biomedicines12081777. Biomedicines. 2024. PMID: 39200241 Free PMC article. Review.
References
-
- Konturek PC, Konturek JW, Czesnikiewicz-Guzik M, Brzozowski T, Sito E, Konturek SJ. Neuro-hormonal control of food intake: basic mechanisms and clinical implications. J Physiol Pharmacol. 2005;56(Suppl 6):5–25. - PubMed
-
- Benelam B. Satiation, satiety and their effects on eating behaviour. 2009;34(2):126–73.
-
- Berthoud HR, Sutton GM, Townsend RL, Patterson LM, Zheng H. Brainstem mechanisms integrating gut-derived satiety signals and descending forebrain information in the control of meal size. Physiol Behav. 2006;89(4):517–24. - PubMed
-
- McMinn JE, Baskin DG, Schwartz MW. Neuroendocrine mechanisms regulating food intake and body weight. Obes Rev. 2000;1(1):37–46. - PubMed

