Molecular diameters of rarefied gases
- PMID: 35136099
- PMCID: PMC8825840
- DOI: 10.1038/s41598-022-05871-y
Molecular diameters of rarefied gases
Abstract
Molecular diameters are an important property of gases for numerous scientific and technical disciplines. Different measurement techniques for these diameters exist, each delivering a characteristic value. Their reliability in describing the flow of rarefied gases, however, has not yet been discussed, especially the case for the transitional range between continuum and ballistic flow. Here, we present a method to describe gas flows in straight channels with arbitrary cross sections for the whole Knudsen range by using a superposition model based on molecular diameters. This model allows us to determine a transition diameter from flow measurement data that paves the way for generalized calculations of gas behaviour under rarefied conditions linking continuum and free molecular regime.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


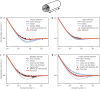
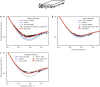


References
-
- Dybkov VI. Reaction diffusion in heterogeneous binary systems. J. Mater. Sci. 1986;21:3078–3084. doi: 10.1007/BF00553339. - DOI
-
- Falkovich G. Fluid Mechanics. Cambridge University Press; 2018.
-
- Baker RW. Membrane Technology and Applications. Wiley; 2012.
-
- van der Bondi A. Waals volumes and radii. J. Phys. Chem. 1964;68:441–451. doi: 10.1021/j100785a001. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

