Glucosamine-6-phosphate N-acetyltransferase gene silencing by parental RNA interference in rice leaf folder, Cnaphalocrocis medinalis (Lepidoptera: Pyralidae)
- PMID: 35136178
- PMCID: PMC8825807
- DOI: 10.1038/s41598-022-06193-9
Glucosamine-6-phosphate N-acetyltransferase gene silencing by parental RNA interference in rice leaf folder, Cnaphalocrocis medinalis (Lepidoptera: Pyralidae)
Abstract
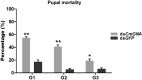
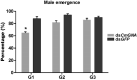
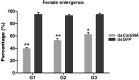
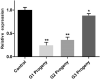
Parental RNAi (pRNAi) is a response of RNA interference in which treated insect pests progenies showed a gene silencing phenotypes. pRNAi of CmGNA gene has been studied in Cnaphalocrocis medinalis via injection. Our results showed significant reduction in ovulation per female that was 26% and 35.26% in G1 and G2 generations, respectively. Significant reduction of hatched eggs per female were observed 23.53% and 45.26% as compared to control in G1-G2 generations, respectively. We also observed the significant variation in the sex ratio between female (40% and 53%) in G1-G2 generations, and in male (65%) in G1 generation as compared to control. Our results also demonstrated the significant larval mortality (63% and 55%) and pupal mortality (55% and 41%), and significant reduction of mRNA expression level in G1 and G2 generations. Our findings have confirmed that effectiveness of pRNAi induced silencing on the CmGNA target gene in G1-G2 generations of C. medinalis. These results suggested the potential role of pRNAi in insect pest resistance management strategies.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures





Similar articles
-
Parental RNA interference (pRNAi) in insects: mechanisms, applications, and challenges in pest management.Mol Biol Rep. 2025 Jun 18;52(1):610. doi: 10.1007/s11033-025-10700-2. Mol Biol Rep. 2025. PMID: 40531353 Review.
-
Knockdown of the glucosamine-6-phosphate N-acetyltransferase gene by RNA interference enhances the virulence of entomopathogenic fungi against rice leaffolder Cnaphalocrocis medinalis.Pestic Biochem Physiol. 2024 Nov;205:106119. doi: 10.1016/j.pestbp.2024.106119. Epub 2024 Sep 4. Pestic Biochem Physiol. 2024. PMID: 39477580
-
Characterization, Knockdown and Parental Effect of Hexokinase Gene of Cnaphalocrocis medinalis (Lepidoptera: Pyralidae) Revealed by RNA Interference.Genes (Basel). 2020 Oct 26;11(11):1258. doi: 10.3390/genes11111258. Genes (Basel). 2020. PMID: 33114530 Free PMC article.
-
Identification and functional analyses of the CmdsRNase5 and CmdsRNase6 genes in rice leaffolder Cnaphalocrocis medinalis.Int J Biol Macromol. 2025 Apr;301:140079. doi: 10.1016/j.ijbiomac.2025.140079. Epub 2025 Jan 23. Int J Biol Macromol. 2025. PMID: 39863202
-
Insecticidal Gene Silencing by RNAi in the Neotropical Region.Neotrop Entomol. 2020 Feb;49(1):1-11. doi: 10.1007/s13744-019-00722-4. Epub 2019 Nov 20. Neotrop Entomol. 2020. PMID: 31749122 Review.
Cited by
-
Parental RNA interference (pRNAi) in insects: mechanisms, applications, and challenges in pest management.Mol Biol Rep. 2025 Jun 18;52(1):610. doi: 10.1007/s11033-025-10700-2. Mol Biol Rep. 2025. PMID: 40531353 Review.
-
Reference Genes for Expression Analysis Using RT-qPCR in Cnaphalocrocis medinalis (Lepidoptera: Pyralidae).Insects. 2022 Nov 13;13(11):1046. doi: 10.3390/insects13111046. Insects. 2022. PMID: 36421949 Free PMC article.
-
Engineering siRNA therapeutics: challenges and strategies.J Nanobiotechnology. 2023 Oct 18;21(1):381. doi: 10.1186/s12951-023-02147-z. J Nanobiotechnology. 2023. PMID: 37848888 Free PMC article. Review.
References
-
- Muthayya S, Sugimoto JD, Montgomery S, Maberly GF. An overview of global rice production, supply, trade, and consumption. Ann. N. Y. Acad. Sci. 2014;1324(1):7–14. - PubMed
-
- Heinrichs EA, Muniappan R. IPM for tropical crops: Rice. CAB Rev. 2017;12(30):1–30.
-
- Peng S, Khush GS. Four decades of breeding for varietal improvement of irrigated lowland rice in the International Rice Research Institute. Plant Prod. Sci. 2003;6(3):157–164.
-
- Long-ping, Y. U. A. N. (2013). Development of hybrid rice to ensure food security. 水稻科学, 21(1), 1–2.
-
- Horgan FG, Crisol E. Hybrid rice and insect herbivores in Asia. Entomol. Exp. Appl. 2013;148(1):1–19.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

