Physiological and Pathological Relevance of Selective and Nonselective Ca2+ Channels in Skeletal and Cardiac Muscle
- PMID: 35138617
- PMCID: PMC10683374
- DOI: 10.1007/978-981-16-4254-8_11
Physiological and Pathological Relevance of Selective and Nonselective Ca2+ Channels in Skeletal and Cardiac Muscle
Abstract
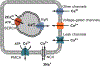
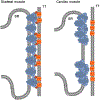
Contraction of the striated muscle is fundamental for human existence. The action of voluntary skeletal muscle enables activities such as breathing, establishing body posture, and diverse body movements. Additionally, highly precise motion empowers communication, artistic expression, and other activities that define everyday human life. The involuntary contraction of striated muscle is the core function of the heart and is essential for blood flow. Several ion channels are important in the transduction of action potentials to cytosolic Ca2+ signals that enable muscle contraction; however, other ion channels are involved in the progression of muscle pathologies that can impair normal life or threaten it. This chapter describes types of selective and nonselective Ca2+ permeable ion channels expressed in the striated muscle, their participation in different aspects of muscle excitation and contraction, and their relevance to the progression of some pathological states.
Keywords: Calcium; Dystrophy; Hypertrophy; Malignant hyperthermia; Orai1; Resting Ca2+ entry; Ryanodine receptor; STIM1; Store-operated Ca2+ entry; TRPC; Voltage-gated calcium channel.
© 2021. Springer Nature Singapore Pte Ltd.
Figures

Similar articles
-
TRPC Channels in the SOCE Scenario.Cells. 2020 Jan 5;9(1):126. doi: 10.3390/cells9010126. Cells. 2020. PMID: 31948094 Free PMC article. Review.
-
Intricate interaction between store-operated calcium entry and calcium-activated chloride channels in pulmonary artery smooth muscle cells.Adv Exp Med Biol. 2010;661:31-55. doi: 10.1007/978-1-60761-500-2_3. Adv Exp Med Biol. 2010. PMID: 20204722
-
Mechanistic insights into store-operated Ca2+ entry during excitation-contraction coupling in skeletal muscle.Biochim Biophys Acta Mol Cell Res. 2019 Jul;1866(7):1239-1248. doi: 10.1016/j.bbamcr.2019.02.014. Epub 2019 Feb 27. Biochim Biophys Acta Mol Cell Res. 2019. PMID: 30825472
-
Evidence that Orai1 does not contribute to store-operated TRPC1 channels in vascular smooth muscle cells.Channels (Austin). 2017 Jul 4;11(4):329-339. doi: 10.1080/19336950.2017.1303025. Epub 2017 Mar 16. Channels (Austin). 2017. PMID: 28301277 Free PMC article.
-
Stim, ORAI and TRPC channels in the control of calcium entry signals in smooth muscle.Clin Exp Pharmacol Physiol. 2008 Sep;35(9):1127-33. doi: 10.1111/j.1440-1681.2008.05018.x. Clin Exp Pharmacol Physiol. 2008. PMID: 18782202 Free PMC article. Review.
Cited by
-
Dantrolene inhibition of ryanodine receptor 1 carrying the severe malignant hyperthermia mutation Y522S visualized by cryo-EM.Structure. 2025 Feb 6;33(2):338-348.e4. doi: 10.1016/j.str.2024.11.018. Epub 2024 Dec 20. Structure. 2025. PMID: 39708816
References
-
- Berridge MJ. Elementary and global aspects of calcium signalling. J Exp Biol. 1997;200(Pt 2):315–9. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

