Surface Ig variable domain glycosylation affects autoantigen binding and acts as threshold for human autoreactive B cell activation
- PMID: 35138894
- PMCID: PMC8827743
- DOI: 10.1126/sciadv.abm1759
Surface Ig variable domain glycosylation affects autoantigen binding and acts as threshold for human autoreactive B cell activation
Abstract
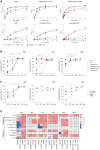
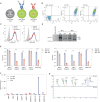
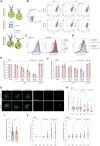
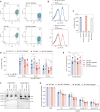
The hallmark autoantibodies in rheumatoid arthritis are characterized by variable domain glycans (VDGs). Their abundant occurrence results from the selective introduction of N-linked glycosylation sites during somatic hypermutation, and their presence is predictive for disease development. However, the functional consequences of VDGs on autoreactive B cells remain elusive. Combining crystallography, glycobiology, and functional B cell assays allowed us to dissect key characteristics of VDGs on human B cell biology. Crystal structures showed that VDGs are positioned in the vicinity of the antigen-binding pocket, and dynamic modeling combined with binding assays elucidated their impact on binding. We found that VDG-expressing B cell receptors stay longer on the B cell surface and that VDGs enhance B cell activation. These results provide a rationale on how the acquisition of VDGs might contribute to the breach of tolerance of autoreactive B cells in a major human autoimmune disease.
Figures





Similar articles
-
A photoaffinity glycan-labeling approach to investigate immunoglobulin glycan-binding partners.Glycobiology. 2023 Oct 29;33(9):732-744. doi: 10.1093/glycob/cwad055. Glycobiology. 2023. PMID: 37498177 Free PMC article.
-
N-Glycosylation Site Analysis of Citrullinated Antigen-Specific B-Cell Receptors Indicates Alternative Selection Pathways During Autoreactive B-Cell Development.Front Immunol. 2019 Sep 4;10:2092. doi: 10.3389/fimmu.2019.02092. eCollection 2019. Front Immunol. 2019. PMID: 31572358 Free PMC article.
-
A Comparison of Immunoglobulin Variable Region N-Linked Glycosylation in Healthy Donors, Autoimmune Disease and Lymphoma.Front Immunol. 2020 Feb 18;11:241. doi: 10.3389/fimmu.2020.00241. eCollection 2020. Front Immunol. 2020. PMID: 32133009 Free PMC article. Review.
-
Adaptive antibody diversification through N-linked glycosylation of the immunoglobulin variable region.Proc Natl Acad Sci U S A. 2018 Feb 20;115(8):1901-1906. doi: 10.1073/pnas.1711720115. Epub 2018 Feb 5. Proc Natl Acad Sci U S A. 2018. PMID: 29432186 Free PMC article.
-
From risk to chronicity: evolution of autoreactive B cell and antibody responses in rheumatoid arthritis.Nat Rev Rheumatol. 2022 Jul;18(7):371-383. doi: 10.1038/s41584-022-00786-4. Epub 2022 May 23. Nat Rev Rheumatol. 2022. PMID: 35606567 Review.
Cited by
-
The CH1 domain influences the expression and antigen sensing of the HIV-specific CH31 IgM-BCR and IgG-BCR.Proc Natl Acad Sci U S A. 2024 Jul 30;121(31):e2404728121. doi: 10.1073/pnas.2404728121. Epub 2024 Jul 23. Proc Natl Acad Sci U S A. 2024. PMID: 39042672 Free PMC article.
-
Effect of posttranslational modifications and subclass on IgG activity: from immunity to immunotherapy.Nat Immunol. 2023 Aug;24(8):1244-1255. doi: 10.1038/s41590-023-01544-8. Epub 2023 Jul 6. Nat Immunol. 2023. PMID: 37414906 Review.
-
Altered IgG N-Glycosylation at Onset of Type 1 Diabetes in Children Is Predominantly Driven by Changes in the Fab N-Glycans.Biomedicines. 2025 May 15;13(5):1206. doi: 10.3390/biomedicines13051206. Biomedicines. 2025. PMID: 40427033 Free PMC article.
-
Distinct mucosal endotypes as initiators and drivers of rheumatoid arthritis.Nat Rev Rheumatol. 2024 Oct;20(10):601-613. doi: 10.1038/s41584-024-01154-0. Epub 2024 Sep 9. Nat Rev Rheumatol. 2024. PMID: 39251771 Review.
-
A photoaffinity glycan-labeling approach to investigate immunoglobulin glycan-binding partners.Glycobiology. 2023 Oct 29;33(9):732-744. doi: 10.1093/glycob/cwad055. Glycobiology. 2023. PMID: 37498177 Free PMC article.
References
-
- Hampe C. S., B cells in autoimmune diseases. Scientifica 2012, 1–18 (2012).
-
- Erikson J., Radic M. Z., Camper S. A., Hardy R. R., Carmack C., Weigert M., Expression of anti-DNA immunoglobulin transgenes in non-autoimmune mice. Nature 349, 331–334 (1991). - PubMed
-
- Goodnow C. C., Crosbie J., Adelstein S., Lavoie T. B., Smith-Gill S. J., Brink R. A., Pritchard-Briscoe H., Wotherspoon J. S., Loblay R. H., Raphael K., Trent R. J., Basten A., Altered immunoglobulin expression and functional silencing of self-reactive B lymphocytes in transgenic mice. Nature 334, 676–682 (1988). - PubMed
-
- Hartley S. B., Crosbie J., Brink R., Kantor A. B., Basten A., Goodnow C. C., Elimination from peripheral lymphoid tissues of self-reactive B lymphocytes recognizing membrane-bound antigens. Nature 353, 765–769 (1991). - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

