Combination therapy for pancreatic cancer: anti-PD-(L)1-based strategy
- PMID: 35139879
- PMCID: PMC8827285
- DOI: 10.1186/s13046-022-02273-w
Combination therapy for pancreatic cancer: anti-PD-(L)1-based strategy
Abstract
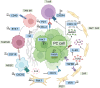
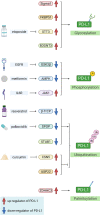
Mortality associated with pancreatic cancer is among the highest of all malignancies, with a 5-year overall survival of 5-10%. Immunotherapy, represented by the blocking antibodies against programmed cell death protein 1 or its ligand 1 (anti-PD-(L)1), has achieved remarkable success in a number of malignancies. However, due to the immune-suppressive tumor microenvironment, the therapeutic efficacy of anti-PD-(L)1 in pancreatic cancer is far from expectation. To address such a fundamental issue, chemotherapy, radiotherapy, targeted therapy and even immunotherapy itself, have individually been attempted to combine with anti-PD-(L)1 in preclinical and clinical investigation. This review, with a particular focus on pancreatic cancer therapy, collects current anti-PD-(L)1-based combination strategy, highlights potential adverse effects of accumulative combination, and further points out future direction in optimization of combination, including targeting post-translational modification of PD-(L)1 and improving precision of treatment.
Keywords: Combinational therapy; Immune checkpoint inhibitor; PD-1; PD-L1; Pancreatic cancer; Post-translational modification; Precision therapy; Systematic treatment.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
References
-
- Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. - PubMed
-
- Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2022. CA Cancer J Clin. 2022;72(1):7–33. - PubMed
-
- Rahib L, Smith BD, Aizenberg R, Rosenzweig AB, Fleshman JM, Matrisian LM. Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 2014;74(11):2913–2921. - PubMed
-
- Mizrahi JD, Surana R, Valle JW, Shroff RT. Pancreatic cancer. Lancet. 2020;395(10242):2008–2020. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

