IL-17 is a Potential Therapeutic Target in a Rodent Model of Otitis Media with Effusion
- PMID: 35140496
- PMCID: PMC8818970
- DOI: 10.2147/JIR.S338598
IL-17 is a Potential Therapeutic Target in a Rodent Model of Otitis Media with Effusion
Abstract
Background: Otitis media with effusion (OME) is a non-suppurative inflammation of the middle ear that is characterized by middle ear effusion and hearing loss. However, the mechanisms of OME are not fully understood. The aim of this study was to determine the function and the mechanism of the IL-17 cytokine in the pathogenesis of OME and to investigate IL-17 as a potential strategy for the treatment of OME.
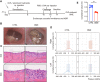
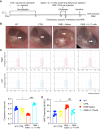
Methods: In this study, the OME rat model was induced by ovalbumin (OVA) as previously described. The severity of OME was determined with an oto-endoscope, by histochemical analysis, and by acoustic immittance. Kyoto Encyclopedia of Genes and Genomes (KEGG) analysis of RNA-sequencing (RNA-seq) data was carried out to analyze the signaling pathways related to the pathogenesis of OME, which indicated that IL-17 is involved in OME. The anti-IL-17A monoclonal antibody was administrated by nasal drip to block IL-17 to treat OME in the rat model. The rats were finally injected intraperitoneally with the inhibitor of Notch signaling pathway to study the mechanisms of IL-17-induced inflammation. Serum and lavage fluid were collected for the detection of related cytokines, and middle ear tissue was collected for Western blot, quantitative real-time PCR (qRT-PCR), and immunohistochemical and immunofluorescence analysis.
Results: KEGG analysis of RNA-seq data suggested that the IL-17 signaling pathway might be involved in the onset of OME. IL-17 expression was confirmed to be increased in both the serum and the middle ear of the rat model. The monoclonal antibody against IL-17 neutralized IL-17, inhibited the inflammation in the middle ear, and reduced the overall severity of OME in vivo. Furthermore, the Notch signaling pathway was activated upon IL-17 upregulation in OME and was suppressed by IL-17 blockage. However, there was no change in IL-17 expression after Notch inhibitor treatment, which reduced the severity of OME in the rat middle ear.
Conclusion: IL-17 plays a key role in the pathogenesis of the OVA-induced OME rat model. IL-17 induced inflammatory responses via the Notch signaling pathway and targeting IL-17 might be an effective approach for OME therapy.
Keywords: IL-17; anti-IL-17A monoclonal antibody; notch; otitis media with effusion.
© 2022 Zhang et al.
Conflict of interest statement
The authors declare that no conflict of interest exists.
Figures




References
LinkOut - more resources
Full Text Sources

