SARS-CoV-2 antibodies protect against reinfection for at least 6 months in a multicentre seroepidemiological workplace cohort
- PMID: 35143473
- PMCID: PMC8865659
- DOI: 10.1371/journal.pbio.3001531
SARS-CoV-2 antibodies protect against reinfection for at least 6 months in a multicentre seroepidemiological workplace cohort
Abstract
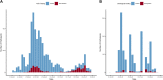
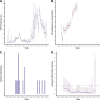
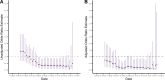
Identifying the potential for SARS-CoV-2 reinfection is crucial for understanding possible long-term epidemic dynamics. We analysed longitudinal PCR and serological testing data from a prospective cohort of 4,411 United States employees in 4 states between April 2020 and February 2021. We conducted a multivariable logistic regression investigating the association between baseline serological status and subsequent PCR test result in order to calculate an odds ratio for reinfection. We estimated an odds ratio for reinfection ranging from 0.14 (95% CI: 0.019 to 0.63) to 0.28 (95% CI: 0.05 to 1.1), implying that the presence of SARS-CoV-2 antibodies at baseline is associated with around 72% to 86% reduced odds of a subsequent PCR positive test based on our point estimates. This suggests that primary infection with SARS-CoV-2 provides protection against reinfection in the majority of individuals, at least over a 6-month time period. We also highlight 2 major sources of bias and uncertainty to be considered when estimating the relative risk of reinfection, confounders and the choice of baseline time point, and show how to account for both in reinfection analysis.
Conflict of interest statement
I have read the journal’s policy and the authors of this manuscript have the following competing interests: GA is a founder of Seromyx Systems Inc., a company developing platform technology that describes the antibody immune response. GA’s interests were reviewed and are managed by Massachusetts General Hospital in accordance with their conflict-of-interest policies. MJG, SB, DD, YH, JR, EP, BM, ASM, and ERM are employees of Space Exploration Technologies Corp. All other authors have declared that no conflict of interest exists.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
- U19 AI135995/AI/NIAID NIH HHS/United States
- MR/V027956/1/MRC_/Medical Research Council/United Kingdom
- U01 GH002238/GH/CGH CDC HHS/United States
- R01 AI042790/AI/NIAID NIH HHS/United States
- R01 AI146785/AI/NIAID NIH HHS/United States
- 206250/Z/17/Z/WT_/Wellcome Trust/United Kingdom
- R01 NS110424/NS/NINDS NIH HHS/United States
- MC_PC_19065/MRC_/Medical Research Council/United Kingdom
- WT_/Wellcome Trust/United Kingdom
- MR/N013638/1/MRC_/Medical Research Council/United Kingdom
- U01 CA260476/CA/NCI NIH HHS/United States
- R37 AI080289/AI/NIAID NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

