Effect of Intra-arterial Alteplase vs Placebo Following Successful Thrombectomy on Functional Outcomes in Patients With Large Vessel Occlusion Acute Ischemic Stroke: The CHOICE Randomized Clinical Trial
- PMID: 35143603
- PMCID: PMC8832304
- DOI: 10.1001/jama.2022.1645
Effect of Intra-arterial Alteplase vs Placebo Following Successful Thrombectomy on Functional Outcomes in Patients With Large Vessel Occlusion Acute Ischemic Stroke: The CHOICE Randomized Clinical Trial
Abstract
Importance: It is estimated that only 27% of patients with acute ischemic stroke and large vessel occlusion who undergo successful reperfusion after mechanical thrombectomy are disability free at 90 days. An incomplete microcirculatory reperfusion might contribute to these suboptimal clinical benefits.
Objective: To investigate whether treatment with adjunct intra-arterial alteplase after thrombectomy improves outcomes following reperfusion.
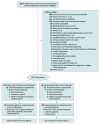
Design, setting, and participants: Phase 2b randomized, double-blind, placebo-controlled trial performed from December 2018 through May 2021 in 7 stroke centers in Catalonia, Spain. The study included 121 patients with large vessel occlusion acute ischemic stroke treated with thrombectomy within 24 hours after stroke onset and with an expanded Treatment in Cerebral Ischemia angiographic score of 2b50 to 3.
Interventions: Participants were randomized to receive intra-arterial alteplase (0.225 mg/kg; maximum dose, 22.5 mg) infused over 15 to 30 minutes (n = 61) or placebo (n = 52).
Main outcomes and measures: The primary outcome was the difference in proportion of patients achieving a score of 0 or 1 on the 90-day modified Rankin Scale (range, 0 [no symptoms] to 6 [death]) in all patients treated as randomized. Safety outcomes included rate of symptomatic intracranial hemorrhage and death.
Results: The study was terminated early for inability to maintain placebo availability and enrollment rate because of the COVID-19 pandemic. Of 1825 patients with acute ischemic stroke treated with thrombectomy at the 7 study sites, 748 (41%) patients fulfilled the angiographic criteria, 121 (7%) patients were randomized (mean age, 70.6 [SD, 13.7] years; 57 women [47%]), and 113 (6%) were treated as randomized. The proportion of participants with a modified Rankin Scale score of 0 or 1 at 90 days was 59.0% (36/61) with alteplase and 40.4% (21/52) with placebo (adjusted risk difference, 18.4%; 95% CI, 0.3%-36.4%; P = .047). The proportion of patients with symptomatic intracranial hemorrhage within 24 hours was 0% with alteplase and 3.8% with placebo (risk difference, -3.8%; 95% CI, -13.2% to 2.5%). Ninety-day mortality was 8% with alteplase and 15% with placebo (risk difference, -7.2%; 95% CI, -19.2% to 4.8%).
Conclusions and relevance: Among patients with large vessel occlusion acute ischemic stroke and successful reperfusion following thrombectomy, the use of adjunct intra-arterial alteplase compared with placebo resulted in a greater likelihood of excellent neurological outcome at 90 days. However, because of study limitations, these findings should be interpreted as preliminary and require replication.
Trial registration: ClinicalTrials.gov Identifier: NCT03876119; EudraCT Number: 2018-002195-40.
Conflict of interest statement
Figures

Comment in
-
Intra-arterial Thrombolysis to Target Occlusions in Distal Arteries and the Microcirculation.JAMA. 2022 Mar 1;327(9):821-823. doi: 10.1001/jama.2021.25014. JAMA. 2022. PMID: 35143600 No abstract available.
-
Intra-arterial Alteplase vs Placebo After Successful Thrombectomy and Functional Outcomes in Patients With Large Vessel Occlusion Acute Ischemic Stroke.JAMA. 2022 Jun 28;327(24):2455-2456. doi: 10.1001/jama.2022.7427. JAMA. 2022. PMID: 35763003 No abstract available.
References
-
- Powers WJ, Rabinstein AA, Ackerson T, et al. Guidelines for the early management of patients with acute ischemic stroke: 2019 update to the 2018 guidelines for the early management of acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2019;50(12):e344-e418. doi: 10.1161/STR.0000000000000211 - DOI - PubMed
-
- Zaidat OO, Yoo AJ, Khatri P, et al. ; Cerebral Angiographic Revascularization Grading Collaborators; STIR Revascularization Working Group; STIR Thrombolysis in Cerebral Infarction Task Force . Recommendations on angiographic revascularization grading standards for acute ischemic stroke: a consensus statement. Stroke. 2013;44(9):2650-2663. doi: 10.1161/STROKEAHA.113.001972 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

