Molecular origin of somatostatin-positive neuron vulnerability
- PMID: 35145229
- PMCID: PMC9133093
- DOI: 10.1038/s41380-022-01463-4
Molecular origin of somatostatin-positive neuron vulnerability
Abstract
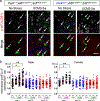
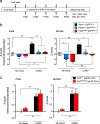
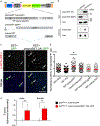
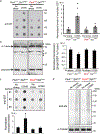
Reduced somatostatin (SST) and dysfunction of SST-positive (SST+) neurons are hallmarks of neurological disorders and associated with mood disturbances, but the molecular origin of SST+ neuron vulnerability is unknown. Using chronic psychosocial stress as a paradigm to induce elevated behavioral emotionality in rodents, we report a selective vulnerability of SST+ neurons through exacerbated unfolded protein response (UPR) of the endoplasmic reticulum (ER), or ER stress, in the prefrontal cortex. We next show that genetically suppressing ER stress in SST+ neurons, but not in pyramidal neurons, normalized behavioral emotionality induced by psychosocial stress. In search for intrinsic factors mediating SST+ neuron vulnerability, we found that the forced expression of the SST precursor protein (preproSST) in SST+ neurons, mimicking psychosocial stress-induced early proteomic changes, induces ER stress, whereas mature SST or processing-incompetent preproSST does not. Biochemical analyses further show that psychosocial stress induces SST protein aggregation under elevated ER stress conditions. These results demonstrate that SST processing in the ER is a SST+ neuron-intrinsic vulnerability factor under conditions of sustained or over-activated UPR, hence negatively impacting SST+ neuron functions. Combined with observations in major medical illness, such as diabetes, where excess ER processing of preproinsulin similarly causes ER stress and β cell dysfunction, this suggests a universal mechanism for proteinopathy that is induced by excess processing of native endogenous proteins, playing critical pathophysiological roles that extend to neuropsychiatric disorders.
© 2022. The Author(s), under exclusive licence to Springer Nature Limited.
Conflict of interest statement
Conflict of Interests
The authors declare no competing interests.
Figures

Similar articles
-
Somatostatin, neuronal vulnerability and behavioral emotionality.Mol Psychiatry. 2015 Mar;20(3):377-87. doi: 10.1038/mp.2014.184. Epub 2015 Jan 20. Mol Psychiatry. 2015. PMID: 25600109 Free PMC article.
-
Opposing effects of acute versus chronic blockade of frontal cortex somatostatin-positive inhibitory neurons on behavioral emotionality in mice.Neuropsychopharmacology. 2014 Aug;39(9):2252-62. doi: 10.1038/npp.2014.76. Epub 2014 Apr 1. Neuropsychopharmacology. 2014. PMID: 24690741 Free PMC article.
-
Behavioral Deficits Induced by Somatostatin-Positive GABA Neuron Silencing Are Rescued by Alpha 5 GABA-A Receptor Potentiation.Int J Neuropsychopharmacol. 2021 Jul 14;24(6):505-518. doi: 10.1093/ijnp/pyab002. Int J Neuropsychopharmacol. 2021. PMID: 33438026 Free PMC article.
-
Molecular Mechanisms of ER Stress and UPR in the Pathogenesis of Alzheimer's Disease.Mol Neurobiol. 2020 Jul;57(7):2902-2919. doi: 10.1007/s12035-020-01929-y. Epub 2020 May 19. Mol Neurobiol. 2020. PMID: 32430843 Review.
-
The Role of Endoplasmic Reticulum Stress in Differentiation of Cells of Mesenchymal Origin.Biochemistry (Mosc). 2022 Sep;87(9):916-931. doi: 10.1134/S000629792209005X. Biochemistry (Mosc). 2022. PMID: 36180988 Free PMC article. Review.
Cited by
-
Transcriptome signatures of the medial prefrontal cortex underlying GABAergic control of resilience to chronic stress exposure.bioRxiv [Preprint]. 2024 Jul 10:2024.07.10.602959. doi: 10.1101/2024.07.10.602959. bioRxiv. 2024. Update in: Mol Psychiatry. 2025 May;30(5):2197-2209. doi: 10.1038/s41380-024-02832-x. PMID: 39026878 Free PMC article. Updated. Preprint.
-
GABAA receptors as targets for treating affective and cognitive symptoms of depression.Trends Pharmacol Sci. 2023 Sep;44(9):586-600. doi: 10.1016/j.tips.2023.06.009. Epub 2023 Aug 3. Trends Pharmacol Sci. 2023. PMID: 37543478 Free PMC article. Review.
-
Inhibitory gamma-aminobutyric acidergic neurons in the anterior cingulate cortex participate in the comorbidity of pain and emotion.Neural Regen Res. 2025 Oct 1;20(10):2838-2854. doi: 10.4103/NRR.NRR-D-24-00429. Epub 2024 Sep 24. Neural Regen Res. 2025. PMID: 39314159 Free PMC article.
-
Somatostatin interneurons and the pathogenesis of Alzheimer's disease.Neural Regen Res. 2026 Mar 1;21(3):1128-1129. doi: 10.4103/NRR.NRR-D-24-01277. Epub 2025 Feb 24. Neural Regen Res. 2026. PMID: 39995091 Free PMC article. No abstract available.
-
Selective inhibition of somatostatin-positive dentate hilar interneurons induces age-related cellular changes and cognitive dysfunction.PNAS Nexus. 2023 Apr 13;2(5):pgad134. doi: 10.1093/pnasnexus/pgad134. eCollection 2023 May. PNAS Nexus. 2023. PMID: 37168673 Free PMC article.
References
-
- Epelbaum J, Guillou J-L, Gastambide F, Hoyer D, Duron E, Viollet C. Somatostatin, Alzheimer’s disease and cognition: an old story coming of age? Prog Neurobiol 2009; 89(2): 153–161. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

