Serious events following COVID-19 vaccination with ChAdOx1 nCoV-19 vaccine (Vaxzevria): A short case series from Iran
- PMID: 35145690
- PMCID: PMC8818285
- DOI: 10.1002/ccr3.5390
Serious events following COVID-19 vaccination with ChAdOx1 nCoV-19 vaccine (Vaxzevria): A short case series from Iran
Abstract
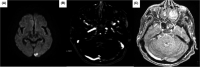
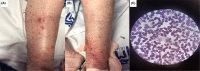
In 2020, the SARS-COV-2 disease (COVID-19) imposed huge challenges on the health, economic, and political systems, and by the end of the year, hope had been born with the release of COVID-19 vaccines aimed at bringing the pandemic to an end. However, the COVID-19 vaccination programs have sparked several concerns and ongoing debates over safety issues. Here, we presented three cases of patients with serious adverse events, encephalopathy, vaccine-induced thrombotic thrombocytopenia, and leukocytoclastic vasculitis, after receiving the ChAdOx1 nCoV-19 vaccine. Therefore, it is critical to investigate and report the occurrence of adverse reactions following vaccination, particularly serious ones, as it contributes to the growing body of research and assists clinicians in better diagnosing and managing them.
Keywords: COVID‐19 vaccination; ChAdOx1 nCoV‐19 vaccine; Leukocytoclastic Vasculitis due to vaccination; adverse effects; postvaccination encephalopathy; vaccine‐induced thrombotic thrombocytopenia.
© 2022 The Authors. Clinical Case Reports published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors state no conflict of interest that could negatively affect the study.
Figures
Similar articles
-
[Intracerebral haemorrhage twelve days after vaccination with ChAdOx1 nCoV-19].Ugeskr Laeger. 2021 Aug 30;183(35):V05210425. Ugeskr Laeger. 2021. PMID: 34477089 Danish.
-
Case Report: Cytomegalovirus Reactivation and Pericarditis Following ChAdOx1 nCoV-19 Vaccination Against SARS-CoV-2.Front Immunol. 2022 Jan 18;12:784145. doi: 10.3389/fimmu.2021.784145. eCollection 2021. Front Immunol. 2022. PMID: 35116025 Free PMC article.
-
Takotsubo (stress) cardiomyopathy after ChAdOx1 nCoV-19 vaccination.BMJ Case Rep. 2021 Oct 8;14(10):e246580. doi: 10.1136/bcr-2021-246580. BMJ Case Rep. 2021. PMID: 34625447 Free PMC article.
-
Possible Risk of Thrombotic Events following Oxford-AstraZeneca COVID-19 Vaccination in Women Receiving Estrogen.Biomed Res Int. 2021 Oct 25;2021:7702863. doi: 10.1155/2021/7702863. eCollection 2021. Biomed Res Int. 2021. PMID: 34734086 Free PMC article. Review.
-
Acute Transverse Myelitis (ATM):Clinical Review of 43 Patients With COVID-19-Associated ATM and 3 Post-Vaccination ATM Serious Adverse Events With the ChAdOx1 nCoV-19 Vaccine (AZD1222).Front Immunol. 2021 Apr 26;12:653786. doi: 10.3389/fimmu.2021.653786. eCollection 2021. Front Immunol. 2021. PMID: 33981305 Free PMC article.
Cited by
-
Cardiac and Neurological Complications Post COVID-19 Vaccination: A Systematic Review of Case Reports and Case Series.Vaccines (Basel). 2024 May 24;12(6):575. doi: 10.3390/vaccines12060575. Vaccines (Basel). 2024. PMID: 38932303 Free PMC article. Review.
-
COVID-19 vaccine-associated vasculitis: A systematic review.SAGE Open Med. 2024 Aug 30;12:20503121241261165. doi: 10.1177/20503121241261165. eCollection 2024. SAGE Open Med. 2024. PMID: 39224893 Free PMC article. Review.
-
COVID-19 Pandemic- Ethical Challenges for Healthcare Workers Practicing in Resource-Limited Settings.Niger Med J. 2022 Sep 16;63(1):1-9. doi: 10.60787/NMJ-63-1-116. eCollection 2022 Jan-Feb. Niger Med J. 2022. PMID: 38798972 Free PMC article. Review.
-
Strategies and safety considerations of booster vaccination in COVID-19.Bosn J Basic Med Sci. 2022 Jun 1;22(3):366-373. doi: 10.17305/bjbms.2021.7082. Bosn J Basic Med Sci. 2022. PMID: 35366790 Free PMC article. Review.
-
A landscape on disorders following different COVID-19 vaccination: a systematic review of Iranian case reports.Eur J Med Res. 2023 Nov 26;28(1):542. doi: 10.1186/s40001-023-01531-7. Eur J Med Res. 2023. PMID: 38008729 Free PMC article.
References
-
- Schultz NH, Sørvoll IH, Michelsen AE, et al. Thrombosis and thrombocytopenia after ChAdOx1 nCoV‐19 vaccination. N Engl J Med. 2021;384(22):2124‐2130. https://pubmed.ncbi.nlm.nih.gov/33835768/ [cited 2021 Aug 30]. - PMC - PubMed
-
- Greinacher A, Thiele T, Warkentin TE, Weisser K, Kyrle PA, Eichinger S. Thrombotic thrombocytopenia after ChAdOx1 nCov‐19 vaccination. N Engl J Med. 2021;384(22):2092‐2101. https://pubmed.ncbi.nlm.nih.gov/33835769/ [cited 2021 Aug 30] - PMC - PubMed
-
- Kadali RAK, Janagama R, Peruru S, Malayala S. Side effects of BNT162b2 mRNA COVID‐19 vaccine: a randomized, cross‐sectional study with detailed self‐reported symptoms from healthcare workers. Int J Infect Dis. 2021;106:376‐381. https://pubmed.ncbi.nlm.nih.gov/33866000/ [cited 2021 Aug 29]. - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

