Electrochemical Cage Activation of Carboranes
- PMID: 35148009
- PMCID: PMC9310615
- DOI: 10.1002/anie.202200323
Electrochemical Cage Activation of Carboranes
Abstract
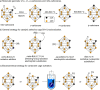
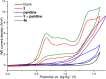
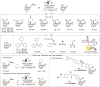
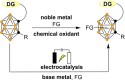
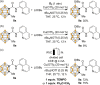
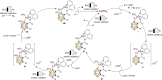
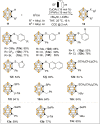
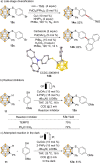
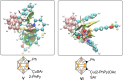
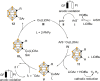
Carboranes are boron-carbon molecular clusters that possess unique properties, such as their icosahedron geometry, high boron content, and delocalized three-dimensional aromaticity. These features render carboranes valuable building blocks for applications in supramolecular design, nanomaterials, optoelectronics, organometallic coordination chemistry, and as boron neutron capture therapy (BNCT) agents. Despite tremendous progress in this field, stoichiometric chemical redox reagents are largely required for the oxidative activation of carborane cages. In this context, electrosyntheses represent an alternative strategy for more sustainable molecular syntheses. It is only in recent few years that considerable progress has been made in electrochemical cage functionalization of carboranes, which are summarized in this Minireview. We anticipate that electrocatalysis will serve as an increasingly powerful stimulus within the current renaissance of carborane electrochemistry.
Keywords: B−H Activation; Cage Activation; Carborane; Electrochemistry.
© 2022 The Authors. Angewandte Chemie International Edition published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





Similar articles
-
A Strategy for Selective Catalytic B-H Functionalization of o-Carboranes.Acc Chem Res. 2021 Nov 2;54(21):4065-4079. doi: 10.1021/acs.accounts.1c00460. Epub 2021 Oct 24. Acc Chem Res. 2021. PMID: 34693715
-
Transition-Metal-Catalyzed Selective Cage B-H Functionalization of o-Carboranes.Chemistry. 2018 Feb 26;24(12):2795-2805. doi: 10.1002/chem.201704937. Epub 2018 Jan 4. Chemistry. 2018. PMID: 29148596
-
Carboranes as unique pharmacophores in antitumor medicinal chemistry.Mol Ther Oncolytics. 2022 Jan 10;24:400-416. doi: 10.1016/j.omto.2022.01.005. eCollection 2022 Mar 17. Mol Ther Oncolytics. 2022. PMID: 35141397 Free PMC article. Review.
-
Site-Selective Functionalization of Carboranes at the Electron-Rich Boron Vertex: Photocatalytic B-C Coupling via a Carboranyl Cage Radical.Angew Chem Int Ed Engl. 2022 Sep 12;61(37):e202205672. doi: 10.1002/anie.202205672. Epub 2022 Jul 8. Angew Chem Int Ed Engl. 2022. PMID: 35670361
-
Controlled functionalization of o-carborane via transition metal catalyzed B-H activation.Chem Soc Rev. 2019 Jul 1;48(13):3660-3673. doi: 10.1039/c9cs00169g. Chem Soc Rev. 2019. PMID: 31090766 Review.
Cited by
-
Site- and enantioselective B-H functionalization of carboranes.Nat Commun. 2025 May 6;16(1):4182. doi: 10.1038/s41467-025-59410-0. Nat Commun. 2025. PMID: 40324985 Free PMC article.
-
Fragile intermediate identification and reactivity elucidation in electrochemical oxidative α-C(sp3)-H functionalization of tertiary amines.Chem Sci. 2023 Mar 18;14(15):4152-4157. doi: 10.1039/d3sc00527e. eCollection 2023 Apr 12. Chem Sci. 2023. PMID: 37063790 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

