High content screening and proteomic analysis identify a kinase inhibitor that rescues pathological phenotypes in a patient-derived model of Parkinson's disease
- PMID: 35149677
- PMCID: PMC8837749
- DOI: 10.1038/s41531-022-00278-y
High content screening and proteomic analysis identify a kinase inhibitor that rescues pathological phenotypes in a patient-derived model of Parkinson's disease
Abstract
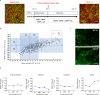
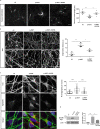
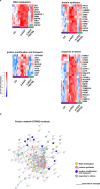
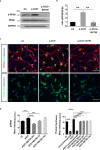
Combining high throughput screening approaches with induced pluripotent stem cell (iPSC)-based disease modeling represents a promising unbiased strategy to identify therapies for neurodegenerative disorders. Here we applied high content imaging on iPSC-derived neurons from patients with familial Parkinson's disease bearing the G209A (p.A53T) α-synuclein (αSyn) mutation and launched a screening campaign on a small kinase inhibitor library. We thus identified the multi-kinase inhibitor BX795 that at a single dose effectively restores disease-associated neurodegenerative phenotypes. Proteomics profiling mapped the molecular pathways underlying the protective effects of BX795, comprising a cohort of 118 protein-mediators of the core biological processes of RNA metabolism, protein synthesis, modification and clearance, and stress response, all linked to the mTORC1 signaling hub. In agreement, expression of human p.A53T-αSyn in neuronal cells affected key components of the mTORC1 pathway resulting in aberrant protein synthesis that was restored in the presence of BX795 with concurrent facilitation of autophagy. Taken together, we have identified a promising small molecule with neuroprotective actions as candidate therapeutic for PD and other protein conformational disorders.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures




References
-
- Olanow CW, Tatton WG. Etiology and pathogenesis of Parkinson’s disease. Annu. Rev. Neurosci. 1999;22:123–144. - PubMed
-
- Pfeiffer RF. Non-motor symptoms in Parkinson’s disease. Parkinsonism Relat. Disord. 2016;22:S119–S122. - PubMed
-
- Gibb W. Idiopathic Parkinson’s disease and the Lewy body disorders. Neuropathol. Appl. Neurobiol. 1986;12:223–234. - PubMed
-
- Lewandowsky, M. H. Handbuch der neurologie: bd. Spezielle Neurologie IV, (J. Spring, 1914).
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

