The radiation adaptive response and priming dose influence: the quantification of the Raper-Yonezawa effect and its three-parameter model for postradiation DNA lesions and mutations
- PMID: 35150289
- PMCID: PMC9021059
- DOI: 10.1007/s00411-022-00963-9
The radiation adaptive response and priming dose influence: the quantification of the Raper-Yonezawa effect and its three-parameter model for postradiation DNA lesions and mutations
Abstract
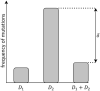
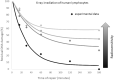
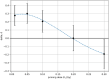
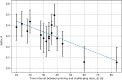
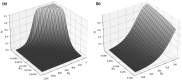
The priming dose effect, called also the Raper-Yonezawa effect or simply the Yonezawa effect, is a special case of the radiation adaptive response phenomenon (radioadaptation), which refers to: (a) faster repair of direct DNA lesions (damage), and (b) DNA mutation frequency reduction after irradiation, by applying a small priming (conditioning) dose prior to the high detrimental (challenging) one. This effect is observed in many (but not all) radiobiological experiments which present the reduction of lesion, mutation or even mortality frequency of the irradiated cells or species. Additionally, the multi-parameter model created by Dr. Yonezawa and collaborators tried to explain it theoretically based on experimental data on the mortality of mice with chronic internal irradiation. The presented paper proposes a new theoretical approach to understanding and explaining the priming dose effect: it starts from the radiation adaptive response theory and moves to the three-parameter model, separately for two previously mentioned situations: creation of fast (lesions) and delayed damage (mutations). The proposed biophysical model was applied to experimental data-lesions in human lymphocytes and chromosomal inversions in mice-and was shown to be able to predict the Yonezawa effect for future investigations. It was also found that the strongest radioadaptation is correlated with the weakest cellular radiosensitivity. Additional discussions were focussed on more general situations where many small priming doses are used.
Keywords: Adaptive response; Cancer physics; Challenging dose; Lymphocytes; Priming dose; Radiation; Radiation biophysics; Radioadaptation; Radiosensitivity; Yonezawa effect.
© 2022. The Author(s).
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures



References
-
- Banzhaf W, Nordin P, Keller R, Francone F. Genetic programming—an introduction. San Francisco: Morgan Kaufmann; 1998.
-
- Bondarenko MA, Zaytseva OV, Trusova VM. Modeling of molecular mechanisms of radiation adaptive response formation. East Eur J Phys. 2021;2:177–188.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

