The Thirty-Day Mortality Rate and Nephrotoxicity Associated With Trough Serum Vancomycin Concentrations During Treatment of Enterococcal Infections: A Propensity Score Matching Analysis
- PMID: 35153743
- PMCID: PMC8831381
- DOI: 10.3389/fphar.2021.773994
The Thirty-Day Mortality Rate and Nephrotoxicity Associated With Trough Serum Vancomycin Concentrations During Treatment of Enterococcal Infections: A Propensity Score Matching Analysis
Abstract
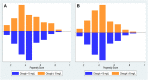
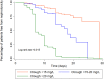
The objective of this study was to evaluate the relationship between vancomycin trough levels in patients with documented enterococcal infections and mortality, clinical outcomes, microbiological outcomes, and nephrotoxicity. We conducted a retrospective cohort study of patients with enterococcus infections who were prescribed vancomycin with therapeutic drug monitoring during January 2010 and December 2019 at Chiang Mai University Hospital (CMUH). The study enrolled 300 participants who met the inclusion criteria and were prescribed vancomycin with therapeutic drug monitoring. The results of this study showed that, after propensity score matching, a vancomycin trough of ≥15 mg/L was associated with significant differences in 30-days mortality compared to a vancomycin trough of <15 mg/L (aHR: 0.41, 95% CI: 0.21-0.82; p = 0.011). Likewise, a vancomycin trough of ≥15 mg/L was associated with significant differences in the clinical response (aHR: 0.49, 95% CI: 0.26-0.94; p = 0.032), microbiological response (aHR: 0.32, 95% CI: 0.12-0.87; p = 0.025) and nephrotoxicity (aHR: 3.17, 95% CI: 1.39-7.23; p = 0.006), compared with a vancomycin trough of <15 mg/L. However, sub-group analysis found that very high trough levels (>20 mg/L) were also associated with a high rate of nephrotoxicity (aHR: 3.55, 95% CI 1.57-8.07, p = 0.002), when compared with a vancomycin trough of <15 mg/L. The target vancomycin trough concentration was ≥15 mg/L and this target can be an optimal alternative to the use of area under the curve (AUC) values for monitoring the treatment of enterococcal infection.
Keywords: enterococcal infections; mortality rate; therapeutic drug monitoring; vancomycin; vancomycin trough levels.
Copyright © 2022 Katip, Okonogi and Oberdorfer.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
-
- CLSI (2015). Performance Standards for Antimicrobial Susceptibility Testing, Twenty-Fifth Informational Supplement. Wayne, PA: Clinical and Laboratory Standards Institute. CLSI doc-ument M100-S25.
-
- CMU antibiotic guidebook (2018). CMU Antibiotic Guidebook. Available at: https://www.scribd.com/document/440356546/Antibiotic-CMU-2557-ORC-shared... (Accessed March 20, 2021).
-
- Corbett A. H., Dana W. J., Fuller M. A. (2016). Drug Information Handbook with International Trade Names Index. 25th ed. (Lexicomp: Wolters Kluwer Inc.), 1353–1356.
LinkOut - more resources
Full Text Sources
Miscellaneous

