Differential Impact of Membrane-Bound and Soluble Forms of the Prognostic Marker Syndecan-1 on the Invasiveness, Migration, Apoptosis, and Proliferation of Cervical Cancer Cells
- PMID: 35155241
- PMCID: PMC8828476
- DOI: 10.3389/fonc.2022.803899
Differential Impact of Membrane-Bound and Soluble Forms of the Prognostic Marker Syndecan-1 on the Invasiveness, Migration, Apoptosis, and Proliferation of Cervical Cancer Cells
Abstract
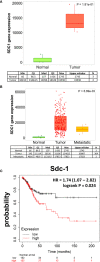
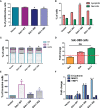
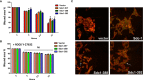
Cervical cancer ranks fourth among the most commonly diagnosed malignant tumors in women worldwide. Previously published evidence suggested a possible connection between the expression of the membrane-bound heparan sulfate proteoglycan syndecan-1 (Sdc-1) and the development of cervical carcinoma. Sdc-1 serves as a matrix receptor and coreceptor for receptor tyrosine kinases and additional signaling pathways. It influences cell proliferation, adhesion, and migration and is seen as a modulator of the tumor microenvironment. Following proteolytic cleavage of its extracellular domain in a process called shedding, Sdc-1 can act as a paracrine effector. The loss of Sdc-1 expression is associated with low differentiation of cervical carcinoma and with an increased rate of lymph node metastases. Here, we analyzed the clinical impact of Sdc-1 expression by analysis of public gene expression datasets and studied the effect of an overexpression of Sdc-1 and its membrane-bound and soluble forms on the malignant properties of the human cervical carcinoma cell line HeLa through functional analysis. For this purpose, the HeLa cells were stably transfected with the control plasmid pcDNA3.1 and three different Sdc-1-DNA constructs,encoding wild-type, permanently membrane-bound, and constitutively soluble Sdc-1. In clinical specimens, Sdc-1 mRNA was more highly expressed in local tumor tissues than in normal and metastatic cervical cancer tissues. Moreover, high Sdc-1 expression correlated with a poor prognosis in Kaplan-Meier survival analysis, suggesting the important role of Sdc-1 in the progression of this type of cancer. In vitro, we found that the soluble, as well as the permanently membrane-bound forms of Sdc-1 modulated the proliferation and the cell cycle, while membrane-bound Sdc1 regulated HeLa cell apoptosis. The overexpression of Sdc-1 and its soluble form increased invasiveness. In vitro scratch/wound healing assay, showed reduced Sdc-1-dependent cell motility which was linked to the Rho-GTPase signaling pathway. In conclusion, in cervical cancer Sdc-1 modulates pathogenetically relevant processes, which depend on the membrane-association of Sdc-1.
Keywords: cervical cancer; metastasis; prognosis; proteoglycan; shedding; syndecan-1.
Copyright © 2022 Hilgers, Ibrahim, Kiesel, Greve, Espinoza-Sánchez and Götte.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
The Heparan Sulfate Proteoglycan Syndecan-1 Triggers Breast Cancer Cell-Induced Coagulability by Induced Expression of Tissue Factor.Cells. 2023 Mar 16;12(6):910. doi: 10.3390/cells12060910. Cells. 2023. PMID: 36980251 Free PMC article.
-
Differential roles for membrane-bound and soluble syndecan-1 (CD138) in breast cancer progression.Carcinogenesis. 2009 Mar;30(3):397-407. doi: 10.1093/carcin/bgp001. Epub 2009 Jan 6. Carcinogenesis. 2009. PMID: 19126645
-
Syndecan-1 Overexpressing Mesothelioma Cells Inhibit Proliferation, Wound Healing, and Tube Formation of Endothelial Cells.Cancers (Basel). 2021 Feb 6;13(4):655. doi: 10.3390/cancers13040655. Cancers (Basel). 2021. PMID: 33562126 Free PMC article.
-
Shed proteoglycans in tumor stroma.Cell Tissue Res. 2016 Sep;365(3):643-55. doi: 10.1007/s00441-016-2452-4. Epub 2016 Jul 1. Cell Tissue Res. 2016. PMID: 27365088 Review.
-
The matrix environmental and cell mechanical properties regulate cell migration and contribute to the invasive phenotype of cancer cells.Rep Prog Phys. 2019 Jun;82(6):064602. doi: 10.1088/1361-6633/ab1628. Epub 2019 Apr 4. Rep Prog Phys. 2019. PMID: 30947151 Review.
Cited by
-
Exostosin 1 Knockdown Induces Chemoresistance in MV3 Melanoma Cells by Upregulating JNK and MEK/ERK Signaling.Int J Mol Sci. 2023 Mar 13;24(6):5452. doi: 10.3390/ijms24065452. Int J Mol Sci. 2023. PMID: 36982528 Free PMC article.
-
Harnessing Human Papillomavirus' Natural Tropism to Target Tumors.Viruses. 2022 Jul 28;14(8):1656. doi: 10.3390/v14081656. Viruses. 2022. PMID: 36016277 Free PMC article. Review.
-
Deciphering the tumor immune microenvironment: single-cell and spatial transcriptomic insights into cervical cancer fibroblasts.J Exp Clin Cancer Res. 2025 Jul 5;44(1):194. doi: 10.1186/s13046-025-03432-5. J Exp Clin Cancer Res. 2025. PMID: 40616092 Free PMC article.
-
Effect of miR-21 in mesenchymal stem cells-derived extracellular vesicles behavior.Stem Cell Res Ther. 2023 Dec 21;14(1):383. doi: 10.1186/s13287-023-03613-z. Stem Cell Res Ther. 2023. PMID: 38129923 Free PMC article.
-
The Heparan Sulfate Proteoglycan Syndecan-1 Triggers Breast Cancer Cell-Induced Coagulability by Induced Expression of Tissue Factor.Cells. 2023 Mar 16;12(6):910. doi: 10.3390/cells12060910. Cells. 2023. PMID: 36980251 Free PMC article.
References
-
- Muñóz–Godínez R, De Lourdes Mora–García M, Weiss–Steider B, Montesinos–Montesinos JJ, Del Carmen Aguilar–Lemarroy A, García–Rocha R, et al. . Detection of CD39 and a Highly Glycosylated Isoform of Soluble CD73 in the Plasma of Patients With Cervical Cancer: Correlation With Disease Progression. Mediators Inflamm (2020) 2020:1678780. doi: 10.1155/2020/1678780 - DOI - PMC - PubMed
-
- García–Rocha R, Moreno–Lafont M, Mora–García ML, Weiss–Steider B, Montesinos JJ, Piña–Sánchez P, et al. . Mesenchymal Stromal Cells Derived From Cervical Cancer Tumors Induce TGF–β1 Expression and IL–10 Expression and Secretion in the Cervical Cancer Cells, Resulting in Protection From Cytotoxic T Cell Activity. Cytokine (2015) 76:382–90. doi: 10.1016/j.cyto.2015.09.001 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

