Apoptosis and Autophagy: Current Understanding in Tick-Pathogen Interactions
- PMID: 35155277
- PMCID: PMC8829008
- DOI: 10.3389/fcimb.2022.784430
Apoptosis and Autophagy: Current Understanding in Tick-Pathogen Interactions
Abstract
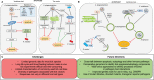
Tick-borne diseases are a significant threat to human and animal health throughout the world. How tick-borne pathogens successfully infect and disseminate in both their vertebrate and invertebrate hosts is only partially understood. Pathogens have evolved several mechanisms to combat host defense systems, and to avoid and modulate host immunity during infection, therefore benefitting their survival and replication. In the host, pathogens trigger responses from innate and adaptive immune systems that recognize and eliminate invaders. Two important innate defenses against pathogens are the programmed cell death pathways of apoptosis and autophagy. This Mini Review surveys the current knowledge of apoptosis and autophagy pathways in tick-pathogen interactions, as well as the strategies evolved by pathogens for their benefit. We then assess the limitations to studying both pathways and discuss their participation in the network of the tick immune system, before highlighting future perspectives in this field. The knowledge gained would significantly enhance our understanding of the defense responses in vector ticks that regulate pathogen infection and burden, and form the foundation for future research to identify novel approaches to the control of tick-borne diseases.
Keywords: Anaplasma; Ehrlichia; Rickettsia; apoptosis; autophagy; cross-talk; intracellular pathogens; tick.
Copyright © 2022 Wang and Cull.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Human pathogens associated with the blacklegged tick Ixodes scapularis: a systematic review.Parasit Vectors. 2016 May 5;9:265. doi: 10.1186/s13071-016-1529-y. Parasit Vectors. 2016. PMID: 27151067 Free PMC article.
-
Behavioral manipulation of Ixodes scapularis by Ehrlichia muris eauclairensis: implications for tick-borne disease transmission.mBio. 2025 Jul 9;16(7):e0075825. doi: 10.1128/mbio.00758-25. Epub 2025 May 27. mBio. 2025. PMID: 40422662 Free PMC article.
-
Detection of pathogens within Ixodid ticks collected from domestic cats across the USA.Parasit Vectors. 2025 Jul 4;18(1):255. doi: 10.1186/s13071-025-06902-z. Parasit Vectors. 2025. PMID: 40615855 Free PMC article.
-
Tick-Borne Diseases in Sub-Saharan Africa: A Systematic Review of Pathogens, Research Focus, and Implications for Public Health.Pathogens. 2024 Aug 17;13(8):697. doi: 10.3390/pathogens13080697. Pathogens. 2024. PMID: 39204297 Free PMC article.
-
Prevalence of tick-borne pathogens in Rhipicephalus species infesting domestic animals in Africa: A systematic review and meta-analysis.Acta Trop. 2023 Oct;246:106994. doi: 10.1016/j.actatropica.2023.106994. Epub 2023 Jul 27. Acta Trop. 2023. PMID: 37516420
Cited by
-
Rickettsia parkeri hypothetical protein RPATATE_1266, a homolog of exopolyphosphatase/guanosine pentaphosphate phosphohydrolase, regulates tick cell apoptosis.Microbiol Spectr. 2025 Aug 5;13(8):e0015125. doi: 10.1128/spectrum.00151-25. Epub 2025 Jul 7. Microbiol Spectr. 2025. PMID: 40621908 Free PMC article.
-
Cuproptosis Cell Death Molecular Events and Pathways to Liver Disease.J Inflamm Res. 2025 Jan 20;18:883-894. doi: 10.2147/JIR.S498340. eCollection 2025. J Inflamm Res. 2025. PMID: 39867947 Free PMC article. Review.
-
Integrative analysis highlights molecular and immune responses of tick Amblyomma americanum to Escherichia coli challenge.Front Cell Infect Microbiol. 2023 Jul 31;13:1236785. doi: 10.3389/fcimb.2023.1236785. eCollection 2023. Front Cell Infect Microbiol. 2023. PMID: 37583446 Free PMC article.
-
The role of autophagy in tick-endosymbiont interactions: insights from Ixodes scapularis and Rickettsia buchneri.Microbiol Spectr. 2024 Jan 11;12(1):e0108623. doi: 10.1128/spectrum.01086-23. Epub 2023 Dec 1. Microbiol Spectr. 2024. PMID: 38038450 Free PMC article.
-
ATF6 enables pathogen infection in ticks by inducing stomatin and altering cholesterol dynamics.Proc Natl Acad Sci U S A. 2025 Jun 24;122(25):e2501045122. doi: 10.1073/pnas.2501045122. Epub 2025 Jun 17. Proc Natl Acad Sci U S A. 2025. PMID: 40526719 Free PMC article.
References
-
- Alberdi P., Mansfield K. L., Manzano-Román R., Cook C., Ayllón N., Villar M., et al. . (2016. b). Tissue-Specific Signatures in the Transcriptional Response to Anaplasma Phagocytophilum Infection of Ixodes Scapularis and Ixodes Ricinus Tick Cell Lines. Front. Cell. Infect. Microbiol. 6, 15. doi: 10.3389/fcimb.2016.00020 - DOI - PMC - PubMed
-
- Ayllón N., Villar M., Galindo R. C., Kocan K. M., Šíma R., López J. A., et al. . (2015). Systems Biology of Tissue-Specific Response to Anaplasma Phagocytophilum Reveals Differentiated Apoptosis in the Tick Vector Ixodes Scapularis. PloS Genet. 11, e1005120. doi: 10.1371/journal.pgen.1005120 - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

