Multiscale model of the physiological control of myocardial perfusion to delineate putative metabolic feedback mechanisms
- PMID: 35156733
- PMCID: PMC9019727
- DOI: 10.1113/JP282237
Multiscale model of the physiological control of myocardial perfusion to delineate putative metabolic feedback mechanisms
Abstract
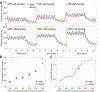
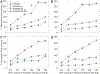
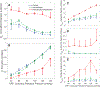
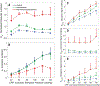
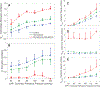
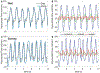
Coronary blood flow is tightly regulated to ensure that myocardial oxygen delivery meets local metabolic demand via the concurrent action of myogenic, neural and metabolic mechanisms. Although several competing hypotheses exist, the specific nature of the local metabolic mechanism(s) remains poorly defined. To gain insights into the viability of putative metabolic feedback mechanisms and into the co-ordinated action of parallel regulatory mechanisms, we applied a multiscale modelling framework to analyse experimental data on coronary pressure, flow and myocardial oxygen delivery in the porcine heart in vivo. The modelling framework integrates a previously established lumped-parameter model of myocardial perfusion used to account for transmural haemodynamic variations and a simple vessel mechanics model used to simulate the vascular tone in each of three myocardial layers. Vascular tone in the resistance vessel mechanics model is governed by input stimuli from the myogenic, metabolic and autonomic control mechanisms. Seven competing formulations of the metabolic feedback mechanism are implemented in the modelling framework, and associated model simulations are compared with experimental data on coronary pressures and flows under a range of experimental conditions designed to interrogate the governing control mechanisms. Analysis identifies a maximally probable metabolic mechanism among the seven tested models, in which production of a metabolic signalling factor is proportional to myocardial oxygen consumption and delivery is proportional to flow. Finally, the identified model is validated based on comparisons of simulations with data on the myocardial perfusion response to conscious exercise that were not used for model identification. KEY POINTS: Although several competing hypotheses exist, we lack knowledge of specific nature of the metabolic mechanism(s) governing regional myocardial perfusion. Moreover, we lack an understanding of how parallel myogenic, adrenergic/autonomic and metabolic mechanisms work together to regulatory oxygen delivery in the beating heart. We have developed a multiscale modelling framework to test competing hypotheses against experimental data on coronary pressure, flow and myocardial oxygen delivery in the porcine heart in vivo. The analysis identifies a maximally probable metabolic mechanism among seven tested models, in which the production of a metabolic signalling factor is proportional to myocardial oxygen consumption and delivery is proportional to flow.
Keywords: coronary blood flow; metabolic control; myocardial perfusion; myogenic control; oxygen transport.
© 2022 The Authors. The Journal of Physiology © 2022 The Physiological Society.
Conflict of interest statement
Competing interests
The authors declare no conflicting interests, financial or otherwise.
Figures


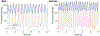
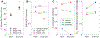

References
-
- Baumgart D, Ehring T, Kowallik P, Guth BD, Krajcar M & Heusch G. (1993). Impact of alpha-adrenergic coronary vasoconstriction on the transmural myocardial blood flow distribution during humoral and neuronal adrenergic activation. Circ Res 73, 869–886. - PubMed
-
- Beau SL, Tolley TK & Saffitz JE. (1993). Heterogeneous transmural distribution of beta-adrenergic receptor subtypes in failing human hearts. Circulation 88, 2501–2509. - PubMed

