A systemic review and meta-analysis on the efficacy and safety of ferumoxytol for anemia in chronic kidney disease patients
- PMID: 35156909
- PMCID: PMC8856040
- DOI: 10.1080/0886022X.2021.2021237
A systemic review and meta-analysis on the efficacy and safety of ferumoxytol for anemia in chronic kidney disease patients
Abstract
Purpose: To evaluate the efficacy of ferumoxytol, relative to conventional iron supplement formulations, on hemoglobin levels, ferritin level, and adverse event incidence in chronic kidney disease patients.
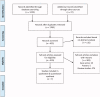
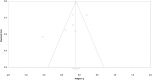
Methods: We performed a systematic search of six academic databases (EMBASE, CENTRAL, Scopus, PubMed, Web of sciences, and MEDLINE), adhering to PRISMA guidelines. We performed a meta-analysis on relevant studies to evaluate the overall influence of ferumoxytol, relative to conventional iron supplement formulations, on hemoglobin levels, ferritin level, and treatment related treatment emergent adverse events (TEAEs) incidence in chronic kidney disease patients.
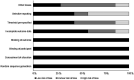
Results: Seven eligible studies were identified from a total of 1397 studies. These studies contained data on 3315 participants with chronic kidney disease (mean age: 59.2 ± 4.6 years). A meta-analysis revealed that ferumoxytol administration had positive effects on hemoglobin levels (Hedge's g statistic: 0.51) and ferritin level (0.88), transferrin saturation (0.39). Besides, we also report reduced incidence of treatment related TEAEs (-0.24) for patients consuming ferumoxytol as compared conventional iron supplement formulations.
Conclusions: This meta-analysis provides preliminary evidence that ferumoxytol use exerts beneficial effects on the overall hematological outcomes in patients with chronic kidney disease. This study also reports improved treatment related safety profile for ferumoxytol when compared with conventional iron formulations. The findings from this study can have direct implications in forming best practice guidelines for managing anemia in patients with chronic kidney disease.
Keywords: Chronic kidney disease; adverse event; ferritin; hemodialysis; hemoglobin.
Conflict of interest statement
No potential conflict of interest was reported by the author(s).
Figures




Comment in
-
Letter to the editor regarding 'A systemic review and meta-analysis on the efficacy and safety of ferumoxytol for anemia in chronic kidney disease patients'.Ren Fail. 2023 Dec;45(1):2153065. doi: 10.1080/0886022X.2022.2153065. Ren Fail. 2023. PMID: 36636996 Free PMC article. No abstract available.
References
-
- Johnson RJ, Feehally J, Floege J.. Comprehensive clinical nephrology E-Book. Philadelphia, PA: Elsevier Health Sciences; 2014.
-
- Zhang L, Wang F, Wang L, et al. Prevalence of chronic kidney disease in China: a cross-sectional survey. Lancet. 2012;379(9818):815–822. - PubMed
-
- Robinson BM, Joffe MM, Berns JS, et al. Anemia and mortality in hemodialysis patients: accounting for morbidity and treatment variables updated over time. Kidney Int. 2005;68(5):2323–2330. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
