KRAS as a Modulator of the Inflammatory Tumor Microenvironment: Therapeutic Implications
- PMID: 35159208
- PMCID: PMC8833974
- DOI: 10.3390/cells11030398
KRAS as a Modulator of the Inflammatory Tumor Microenvironment: Therapeutic Implications
Abstract
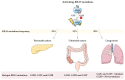
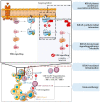
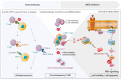
KRAS mutations are one of the most frequent oncogenic mutations of all human cancers, being more prevalent in pancreatic, colorectal, and lung cancers. Intensive efforts have been encouraged in order to understand the effect of KRAS mutations, not only on tumor cells but also on the dynamic network composed by the tumor microenvironment (TME). The relevance of the TME in cancer biology has been increasing due to its impact on the modulation of cancer cell activities, which can dictate the success of tumor progression. Here, we aimed to clarify the pro- and anti-inflammatory role of KRAS mutations over the TME, detailing the context and the signaling pathways involved. In this review, we expect to open new avenues for investigating the potential of KRAS mutations on inflammatory TME modulation, opening a different vision of therapeutic combined approaches to overcome KRAS-associated therapy inefficacy and resistance in cancer.
Keywords: KRAS mutations; colorectal cancer; inflammation; lung cancer; pancreatic cancer; therapy; tumor microenvironment.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Cazzanelli G., Pereira F., Alves S., Francisco R., Azevedo L., Dias Carvalho P., Almeida A., Côrte-Real M., Oliveira M., Lucas C., et al. The Yeast Saccharomyces cerevisiae as a Model for Understanding RAS Proteins and their Role in Human Tumorigenesis. Cells. 2018;7:14. doi: 10.3390/cells7020014. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

