Energy Drinks and Their Acute Effects on Heart Rhythm and Electrocardiographic Time Intervals in Healthy Children and Teenagers: A Randomized Trial
- PMID: 35159306
- PMCID: PMC8834195
- DOI: 10.3390/cells11030498
Energy Drinks and Their Acute Effects on Heart Rhythm and Electrocardiographic Time Intervals in Healthy Children and Teenagers: A Randomized Trial
Abstract
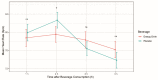
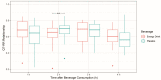
Beyond their effect on blood pressure, the effect of energy drinks on heart rate in children and teenagers has not been evaluated until now. Thus, this study aimed to investigate the acute cardiovascular effects of energy drinks in healthy children and teenagers. Twenty-six children and adolescents (mean age 14.49 years) received a commercially available energy drink (ED) and placebo on two consecutive days based on the maximum caffeine dosage as proposed by the European Food Safety Authority. Heart rhythm and electrocardiographic time intervals were assessed in a prospective, randomized, double-blind, placebo-controlled, crossover clinical study design. ED consumption resulted in a significantly increased number of supraventricular extrasystoles (SVES) compared to the placebo, whereas supraventricular tachycardia or malignant ventricular arrhythmias were not observed. The mean heart rate (HR) was significantly lower following consumption of EDs. In contrast, QTc intervals were not affected by EDs. Being the first of its kind, this trial demonstrates the cardiovascular and rhythmological effects of EDs in minors. Interestingly, EDs were associated with adverse effects on heart rhythm. Whether higher dosages or consumption in children with preexisting conditions may cause potentially harmful disorders was beyond the scope of this pilot study and remains to be determined in future trials. Trial Registration Number (DRKS-ID): DRKS00027580.
Keywords: arrhythmias; caffeine; electrocardiography; energy drinks; extrasystole; heart rate.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Insights I.M. Energy Drink Sales Still on the Rise, Despite Slowdown in Innovation. Nutritional Outlook. Jun 28, 2018.
-
- Zucconi S., Volpato C., Adinolfi F., Gandini E., Gentile E., Loi A., Fioriti L. Gathering Consumption Data on Specific Consumer Groups of Energy Drinks. EFSA Support. Publ. 2013;10:394E. doi: 10.2903/sp.efsa.2013.EN-394. - DOI
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

