Review on Graphene-, Graphene Oxide-, Reduced Graphene Oxide-Based Flexible Composites: From Fabrication to Applications
- PMID: 35160958
- PMCID: PMC8838127
- DOI: 10.3390/ma15031012
Review on Graphene-, Graphene Oxide-, Reduced Graphene Oxide-Based Flexible Composites: From Fabrication to Applications
Abstract
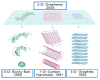
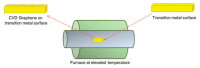
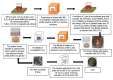
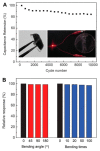
In the new era of modern flexible and bendable technology, graphene-based materials have attracted great attention. The excellent electrical, mechanical, and optical properties of graphene as well as the ease of functionalization of its derivates have enabled graphene to become an attractive candidate for the construction of flexible devices. This paper provides a comprehensive review about the most recent progress in the synthesis and applications of graphene-based composites. Composite materials based on graphene, graphene oxide (GO), and reduced graphene oxide (rGO), as well as conducting polymers, metal matrices, carbon-carbon matrices, and natural fibers have potential application in energy-harvesting systems, clean-energy storage devices, and wearable and portable electronics owing to their superior mechanical strength, conductivity, and extraordinary thermal stability. Additionally, the difficulties and challenges in the current development of graphene are summarized and indicated. This review provides a comprehensive and useful database for further innovation of graphene-based composite materials.
Keywords: composite; flexible devices; graphene; graphene oxide; reduced graphene oxide.
Conflict of interest statement
The authors declare no conflict of interest.
Figures












References
-
- Jalili R., Aboutalebi S.H., Esrafilzadeh D., Shepherd R.L., Chen J., Aminorroaya–Yamini S., Konstantinov K., Minett A.I., Razal J.M., Wallace G.G. Scalable one–step wet–spinning of graphene fibers and yarns from liquid crystalline dispersions of graphene oxide: Towards multifunctional textiles. Adv. Funct. Mater. 2013;23:5345–5354. doi: 10.1002/adfm.201300765. - DOI
-
- Lundstedt A., Papadakis R., Li H., Han Y., Jorner K., Bergman J., Leifer K., Grennberg H., Ottosson H. White–Light Photoassisted Covalent Functionalization of Graphene Using 2–Propanol. Small Methods. 2017;1:1700214. doi: 10.1002/smtd.201700214. - DOI
-
- Li H., Papadakis R., Jafri S.H.M., Thersleff T., Michler J., Ottosson H., Leifer K. Superior adhesion of graphene nanoscrolls. Commun. Phys. 2018;1:44. doi: 10.1038/s42005-018-0043-2. - DOI
-
- Chabot V., Higgins D., Yu A., Xiao X., Chen Z., Zhang J. A review of graphene and graphene oxide sponge: Material synthesis and applications to energy and the environment. Energy Environ. Sci. 2014;7:1564–1596. doi: 10.1039/c3ee43385d. - DOI
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

