Improving Polysaccharide-Based Chitin/Chitosan-Aerogel Materials by Learning from Genetics and Molecular Biology
- PMID: 35160985
- PMCID: PMC8839503
- DOI: 10.3390/ma15031041
Improving Polysaccharide-Based Chitin/Chitosan-Aerogel Materials by Learning from Genetics and Molecular Biology
Abstract
Improved wound healing of burnt skin and skin lesions, as well as medical implants and replacement products, requires the support of synthetical matrices. Yet, producing synthetic biocompatible matrices that exhibit specialized flexibility, stability, and biodegradability is challenging. Synthetic chitin/chitosan matrices may provide the desired advantages for producing specialized grafts but must be modified to improve their properties. Synthetic chitin/chitosan hydrogel and aerogel techniques provide the advantages for improvement with a bioinspired view adapted from the natural molecular toolbox. To this end, animal genetics provide deep knowledge into which molecular key factors decisively influence the properties of natural chitin matrices. The genetically identified proteins and enzymes control chitin matrix assembly, architecture, and degradation. Combining synthetic chitin matrices with critical biological factors may point to the future direction with engineering materials of specific properties for biomedical applications such as burned skin or skin blistering and extensive lesions due to genetic diseases.
Keywords: aerogel; chitin; chitosan; drosophila; insect; materials; obst-A; proteins; skin substitutes; wound healing.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

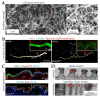

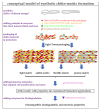
Similar articles
-
Emerging chitin and chitosan nanofibrous materials for biomedical applications.Nanoscale. 2014 Aug 21;6(16):9477-93. doi: 10.1039/c4nr02814g. Nanoscale. 2014. PMID: 25000536 Review.
-
Analysis, Properties, and Applications of Insect-Derived Chitosan: A Sustainable Path to Functional Polysaccharide Materials.Gels. 2025 Apr 15;11(4):291. doi: 10.3390/gels11040291. Gels. 2025. PMID: 40277727 Free PMC article. Review.
-
[The possibilities of using a chitin and chitosan in wounds treatment].Polim Med. 2013 Oct-Dec;43(4):297-302. Polim Med. 2013. PMID: 24596043 Review. Polish.
-
Chitosan functionalized poly(vinyl alcohol) for prospects biomedical and industrial applications: A review.Int J Biol Macromol. 2016 Jun;87:141-54. doi: 10.1016/j.ijbiomac.2016.02.035. Epub 2016 Feb 15. Int J Biol Macromol. 2016. PMID: 26893051 Review.
-
Applications of chitin and chitosan nanofibers in bone regenerative engineering.Carbohydr Polym. 2020 Feb 15;230:115658. doi: 10.1016/j.carbpol.2019.115658. Epub 2019 Nov 23. Carbohydr Polym. 2020. PMID: 31887899 Review.
Cited by
-
A dynamic interplay between chitin synthase and the proteins Expansion/Rebuf reveals that chitin polymerisation and translocation are uncoupled in Drosophila.PLoS Biol. 2023 Jan 23;21(1):e3001978. doi: 10.1371/journal.pbio.3001978. eCollection 2023 Jan. PLoS Biol. 2023. PMID: 36689563 Free PMC article.
-
The Use of Chitosan-Coated Nanovesicles in Repairing Alcohol-Induced Damage of Liver Cells in Mice.Medicina (Kaunas). 2022 Jun 5;58(6):762. doi: 10.3390/medicina58060762. Medicina (Kaunas). 2022. PMID: 35744025 Free PMC article.
-
A review of acaricides and their resistance mechanisms in hard ticks and control alternatives with synergistic agents.Acta Trop. 2025 Jan;261:107519. doi: 10.1016/j.actatropica.2024.107519. Epub 2024 Dec 31. Acta Trop. 2025. PMID: 39746593 Review.
-
Bioinspired Hydrogels as Platforms for Life-Science Applications: Challenges and Opportunities.Polymers (Basel). 2022 Jun 11;14(12):2365. doi: 10.3390/polym14122365. Polymers (Basel). 2022. PMID: 35745941 Free PMC article. Review.
-
Plant polysaccharide-capped nanoparticles: A sustainable approach to modulate gut microbiota and advance functional food applications.Compr Rev Food Sci Food Saf. 2025 Mar;24(2):e70156. doi: 10.1111/1541-4337.70156. Compr Rev Food Sci Food Saf. 2025. PMID: 40052474 Free PMC article. Review.
References
-
- Hong M.-S., Choi G.-M., Kim J., Jang J., Choi B., Kim J.-K., Jeong S., Leem S., Kwon H.-Y., Hwang H.-B., et al. Biomimetic chitin–silk hybrids: An optically transparent structural platform for wearable devices and advanced electronics. Adv. Funct. Mater. 2018;28:1705480. doi: 10.1002/adfm.201705480. - DOI
Publication types
LinkOut - more resources
Full Text Sources

