Critical Determinants in ER-Golgi Trafficking of Enzymes Involved in Glycosylation
- PMID: 35161411
- PMCID: PMC8840164
- DOI: 10.3390/plants11030428
Critical Determinants in ER-Golgi Trafficking of Enzymes Involved in Glycosylation
Abstract
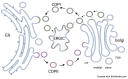
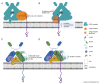
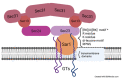
All living cells generate structurally complex and compositionally diverse spectra of glycans and glycoconjugates, critical for organismal evolution, development, functioning, defense, and survival. Glycosyltransferases (GTs) catalyze the glycosylation reaction between activated sugar and acceptor substrate to synthesize a wide variety of glycans. GTs are distributed among more than 130 gene families and are involved in metabolic processes, signal pathways, cell wall polysaccharide biosynthesis, cell development, and growth. Glycosylation mainly takes place in the endoplasmic reticulum (ER) and Golgi, where GTs and glycosidases involved in this process are distributed to different locations of these compartments and sequentially add or cleave various sugars to synthesize the final products of glycosylation. Therefore, delivery of these enzymes to the proper locations, the glycosylation sites, in the cell is essential and involves numerous secretory pathway components. This review presents the current state of knowledge about the mechanisms of protein trafficking between ER and Golgi. It describes what is known about the primary components of protein sorting machinery and trafficking, which are recognition sites on the proteins that are important for their interaction with the critical components of this machinery.
Keywords: COPI and COPII complexes; ER-Golgi trafficking; glycosyltransferases; mechanism of protein sorting; sequences and motifs involved in trafficking.
Conflict of interest statement
The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures
Similar articles
-
Golgi glycosylation.Cold Spring Harb Perspect Biol. 2011 Apr 1;3(4):a005199. doi: 10.1101/cshperspect.a005199. Cold Spring Harb Perspect Biol. 2011. PMID: 21441588 Free PMC article. Review.
-
In tobacco leaf epidermal cells, the integrity of protein export from the endoplasmic reticulum and of ER export sites depends on active COPI machinery.Plant J. 2006 Apr;46(1):95-110. doi: 10.1111/j.1365-313X.2006.02675.x. Plant J. 2006. PMID: 16553898
-
Conserved oligomeric Golgi complex specifically regulates the maintenance of Golgi glycosylation machinery.Glycobiology. 2011 Dec;21(12):1554-69. doi: 10.1093/glycob/cwr028. Epub 2011 Mar 18. Glycobiology. 2011. PMID: 21421995 Free PMC article.
-
The Close Relationship between the Golgi Trafficking Machinery and Protein Glycosylation.Cells. 2020 Dec 10;9(12):2652. doi: 10.3390/cells9122652. Cells. 2020. PMID: 33321764 Free PMC article. Review.
-
Polysaccharide Biosynthesis: Glycosyltransferases and Their Complexes.Front Plant Sci. 2021 Feb 19;12:625307. doi: 10.3389/fpls.2021.625307. eCollection 2021. Front Plant Sci. 2021. PMID: 33679837 Free PMC article.
Cited by
-
Proteomic analysis of hydrogen peroxide-treated human chondrocytes shows endoplasmic reticulum stress, cytoskeleton remodeling, and altered secretome composition.Cell Commun Signal. 2025 Jun 13;23(1):282. doi: 10.1186/s12964-025-02291-z. Cell Commun Signal. 2025. PMID: 40514670 Free PMC article.
-
Mass Spectrometry-Based Glycoproteomic Workflows for Cancer Biomarker Discovery.Technol Cancer Res Treat. 2023 Jan-Dec;22:15330338221148811. doi: 10.1177/15330338221148811. Technol Cancer Res Treat. 2023. PMID: 36740994 Free PMC article. Review.
-
10th Anniversary of Plants-Recent Advances and Further Perspectives.Plants (Basel). 2023 Apr 18;12(8):1696. doi: 10.3390/plants12081696. Plants (Basel). 2023. PMID: 37111918 Free PMC article.
-
Xyloglucan Biosynthesis: From Genes to Proteins and Their Functions.Front Plant Sci. 2022 Jun 2;13:920494. doi: 10.3389/fpls.2022.920494. eCollection 2022. Front Plant Sci. 2022. PMID: 35720558 Free PMC article. Review.
-
Multiprotein Complexes of Plant Glycosyltransferases Involved in Their Function and Trafficking.Plants (Basel). 2025 Jan 24;14(3):350. doi: 10.3390/plants14030350. Plants (Basel). 2025. PMID: 39942912 Free PMC article. Review.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

