Kynurenic Acid and Its Analog SZR104 Exhibit Strong Antiinflammatory Effects and Alter the Intracellular Distribution and Methylation Patterns of H3 Histones in Immunochallenged Microglia-Enriched Cultures of Newborn Rat Brains
- PMID: 35163002
- PMCID: PMC8835130
- DOI: 10.3390/ijms23031079
Kynurenic Acid and Its Analog SZR104 Exhibit Strong Antiinflammatory Effects and Alter the Intracellular Distribution and Methylation Patterns of H3 Histones in Immunochallenged Microglia-Enriched Cultures of Newborn Rat Brains
Abstract
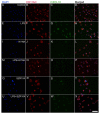
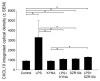
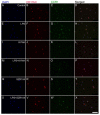
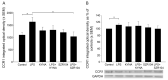
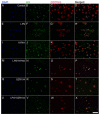
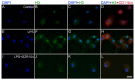
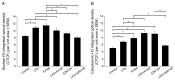
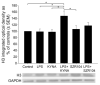
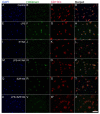
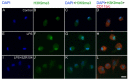
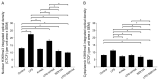
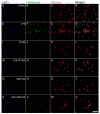
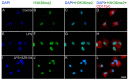
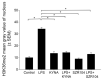
Kynurenic acid (KYNA) is implicated in antiinflammatory processes in the brain through several cellular and molecular targets, among which microglia-related mechanisms are of paramount importance. In this study, we describe the effects of KYNA and one of its analogs, the brain-penetrable SZR104 (N-(2-(dimethylamino)ethyl)-3-(morpholinomethyl)-4-hydroxyquinoline-2-carboxamide), on the intracellular distribution and methylation patterns of histone H3 in immunochallenged microglia cultures. Microglia-enriched secondary cultures made from newborn rat forebrains were immunochallenged with lipopolysaccharide (LPS). The protein levels of selected inflammatory markers C-X-C motif chemokine ligand 10 (CXCL10) and C-C motif chemokine receptor 1 (CCR1), histone H3, and posttranslational modifications of histone H3 lys methylation sites (H3K9me3 and H3K36me2, marks typically associated with opposite effects on gene expression) were analyzed using quantitative fluorescent immunocytochemistry and western blots in control or LPS-treated cultures with or without KYNA or SZR104. KYNA and SZR104 reduced levels of the inflammatory marker proteins CXCL10 and CCR1 after LPS-treatment. Moreover, KYNA and SZR104 favorably affected histone methylation patterns as H3K9me3 and H3K36me2 immunoreactivities, and histone H3 protein levels returned toward control values after LPS treatment. The cytoplasmic translocation of H3K9me3 from the nucleus indicated inflammatory distress, a process that could be inhibited by KYNA and SZR104. Thus, KYNA signaling and metabolism, and especially brain-penetrable KYNA analogs such as SZR104, could be key targets in the pathway that connects chromatin structure and epigenetic mechanisms with functional consequences that affect neuroinflammation and perhaps neurodegeneration.
Keywords: CCR1; CXCL10; H3K36me2; H3K9me3; SZR104; antiinflammation; cytoplasmic histone; kynurenic acid.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures
Similar articles
-
The kynurenic acid analog SZR104 induces cytomorphological changes associated with the anti-inflammatory phenotype in cultured microglia.Sci Rep. 2023 Jul 13;13(1):11328. doi: 10.1038/s41598-023-38107-8. Sci Rep. 2023. PMID: 37443330 Free PMC article.
-
Sensitivity of Rodent Microglia to Kynurenines in Models of Epilepsy and Inflammation In Vivo and In Vitro: Microglia Activation is Inhibited by Kynurenic Acid and the Synthetic Analogue SZR104.Int J Mol Sci. 2020 Dec 7;21(23):9333. doi: 10.3390/ijms21239333. Int J Mol Sci. 2020. PMID: 33297593 Free PMC article.
-
The effect of minocycline on indolamine 2, 3 dioxygenase expression and the levels of kynurenic acid and quinolinic acid in LPS-activated primary rat microglia.Cytokine. 2018 Jul;107:125-129. doi: 10.1016/j.cyto.2017.12.013. Epub 2017 Dec 13. Cytokine. 2018. PMID: 29246652
-
Epigenetic landscape of amphetamine and methamphetamine addiction in rodents.Epigenetics. 2015;10(7):574-80. doi: 10.1080/15592294.2015.1055441. Epigenetics. 2015. PMID: 26023847 Free PMC article. Review.
-
Electrophysiological effects of exogenous and endogenous kynurenic acid in the rat brain: studies in vivo and in vitro.Amino Acids. 2000;19(1):283-97. doi: 10.1007/s007260070060. Amino Acids. 2000. PMID: 11026500 Review.
Cited by
-
The function of previously unappreciated exerkines secreted by muscle in regulation of neurodegenerative diseases.Front Mol Neurosci. 2024 Jan 5;16:1305208. doi: 10.3389/fnmol.2023.1305208. eCollection 2023. Front Mol Neurosci. 2024. PMID: 38249295 Free PMC article. Review.
-
Kynurenine Pathway in Epilepsy: Unraveling Its Role in Glutamate Excitotoxicity, GABAergic Dysregulation, Neuroinflammation, and Mitochondrial Dysfunction.Neurotox Res. 2025 Mar 28;43(2):18. doi: 10.1007/s12640-025-00738-2. Neurotox Res. 2025. PMID: 40153181 Review.
-
The Biology and Biochemistry of Kynurenic Acid, a Potential Nutraceutical with Multiple Biological Effects.Int J Mol Sci. 2024 Aug 21;25(16):9082. doi: 10.3390/ijms25169082. Int J Mol Sci. 2024. PMID: 39201768 Free PMC article. Review.
-
Revolutionizing our understanding of Parkinson's disease: Dr. Heinz Reichmann's pioneering research and future research direction.J Neural Transm (Vienna). 2024 Dec;131(12):1367-1387. doi: 10.1007/s00702-024-02812-z. Epub 2024 Aug 7. J Neural Transm (Vienna). 2024. PMID: 39110245 Free PMC article. Review.
-
Diverse Physiological Roles of Kynurenine Pathway Metabolites: Updated Implications for Health and Disease.Metabolites. 2025 Mar 20;15(3):210. doi: 10.3390/metabo15030210. Metabolites. 2025. PMID: 40137174 Free PMC article. Review.
References
-
- Lajkó N., Kata D., Szabó M., Mátyás A., Dulka K., Földesi I., Fülöp F., Gulya K., Vécsei L., Mihály A. Sensitivity of rodent microglia to kynurenines in models of epilepsy and inflammation in vivo and in vitro: Microglia activation is inhibited by kynurenic acid and the synthetic analogue SZR104. Int. J. Mol. Sci. 2020;21:9333. doi: 10.3390/ijms21239333. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

