Regulation of the Bud Dormancy Development and Release in Micropropagated Rhubarb 'Malinowy'
- PMID: 35163404
- PMCID: PMC8835828
- DOI: 10.3390/ijms23031480
Regulation of the Bud Dormancy Development and Release in Micropropagated Rhubarb 'Malinowy'
Abstract
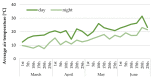
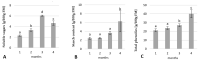
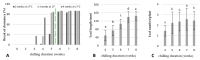
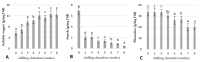
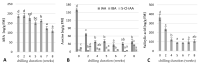
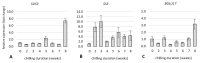
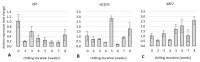
Culinary rhubarb is a vegetable crop, valued for its stalks, very rich in different natural bioactive ingredients. In commercial rhubarb stalk production, the bud dormancy development and release are crucial processes that determine the yields and quality of stalks. To date, reports on rhubarb bud dormancy regulation, however, are lacking. It is known that dormancy status depends on cultivars. The study aimed to determine the dormancy regulation in a valuable selection of rhubarb 'Malinowy'. Changes in carbohydrate, total phenolic, endogenous hormone levels, and gene expression levels during dormancy development and release were studied in micropropagated rhubarb plantlets. Dormancy developed at high temperature (25.5 °C), and long day. Leaf senescence and dying were consistent with a significant increase in starch, total phenolics, ABA, IAA and SA levels. Five weeks of cooling at 4 °C were sufficient to break dormancy, but rhizomes stored for a longer duration showed faster and more uniformity leaf growing, and higher stalk length. No growth response was observed for non-cooled rhizomes. The low temperature activated carbohydrate and hormone metabolism and signalling in the buds. The increased expression of AMY3, BMY3, SUS3, BGLU17, GAMYB genes were consistent with a decrease in starch and increase in soluble sugars levels during dormancy release. Moreover, some genes (ZEP, ABF2, GASA4, GA2OX8) related to ABA and GA metabolism and signal transduction were activated. The relationship between auxin (IAA, IBA, 5-Cl-IAA), and phenolic, including SA levels and dormancy status was also observed.
Keywords: endogenous hormone; gene expression; phenolics; soluble sugars; starch; temperature.
Conflict of interest statement
The authors declare no conflict of interest.
Figures






References
-
- Chao W.S., Foley M.E., Horvath D.P., Anderson J.V. Signalling regulating dormancy in vegetative buds. Int. J. Plant Dev. Biol. 2007;1:49–56.
-
- Podwyszyńska M. The mechanisms of in vitro storage organ formation in ornamental geophytes. Floricult. Ornam. Biotechnol. 2012;6:9–23.
-
- Lang G.A., Early J.D., Martin G.C., Darnell R.L. Endo-, para-, and ecodormancy: Physiological terminology and classification for dormancy research. HortScience. 1987;22:371–377.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

