The Cellular and Subcellular Organization of the Glucosinolate-Myrosinase System against Herbivores and Pathogens
- PMID: 35163500
- PMCID: PMC8836197
- DOI: 10.3390/ijms23031577
The Cellular and Subcellular Organization of the Glucosinolate-Myrosinase System against Herbivores and Pathogens
Abstract
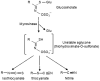
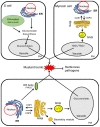
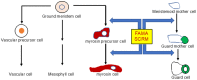
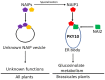
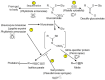
Glucosinolates are an important class of secondary metabolites in Brassicales plants with a critical role in chemical defense. Glucosinolates are chemically inactive but can be hydrolyzed by myrosinases to produce a range of chemically active compounds toxic to herbivores and pathogens, thereby constituting the glucosinolate-myrosinase defense system or the mustard oil bomb. During the evolution, Brassicales plants have developed not only complex biosynthetic pathways for production of a large number of glucosinolate structures but also different classes of myrosinases that differ in catalytic mechanisms and substrate specificity. Studies over the past several decades have made important progress in the understanding of the cellular and subcellular organization of the glucosinolate-myrosinase system for rapid and timely detonation of the mustard oil bomb upon tissue damage after herbivore feeding and pathogen infection. Progress has also been made in understanding the mechanisms that herbivores and pathogens have evolved to counter the mustard oil bomb. In this review, we summarize our current understanding of the function and organization of the glucosinolate-myrosinase system in Brassicales plants and discuss both the progresses and future challenges in addressing this complex defense system as an excellent model for analyzing plant chemical defense.
Keywords: ER body; glucosinolates; mustard bomb; myrosin cells; myrosinases; plant chemical defense.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Insect herbivore counteradaptations to the plant glucosinolate-myrosinase system.Phytochemistry. 2011 Sep;72(13):1566-75. doi: 10.1016/j.phytochem.2011.01.016. Epub 2011 Feb 10. Phytochemistry. 2011. PMID: 21316065 Review.
-
Specialized Vacuoles of Myrosin Cells: Chemical Defense Strategy in Brassicales Plants.Plant Cell Physiol. 2018 Jul 1;59(7):1309-1316. doi: 10.1093/pcp/pcy082. Plant Cell Physiol. 2018. PMID: 29897512 Review.
-
Phyllotreta striolata flea beetles use host plant defense compounds to create their own glucosinolate-myrosinase system.Proc Natl Acad Sci U S A. 2014 May 20;111(20):7349-54. doi: 10.1073/pnas.1321781111. Epub 2014 May 5. Proc Natl Acad Sci U S A. 2014. PMID: 24799680 Free PMC article.
-
Oilseed rape seeds with ablated defence cells of the glucosinolate-myrosinase system. Production and characteristics of double haploid MINELESS plants of Brassica napus L.J Exp Bot. 2011 Oct;62(14):4975-93. doi: 10.1093/jxb/err195. Epub 2011 Jul 21. J Exp Bot. 2011. PMID: 21778185 Free PMC article.
-
Plant defence responses in oilseed rape MINELESS plants after attack by the cabbage moth Mamestra brassicae.J Exp Bot. 2015 Feb;66(2):579-92. doi: 10.1093/jxb/eru490. Epub 2015 Jan 6. J Exp Bot. 2015. PMID: 25563968 Free PMC article.
Cited by
-
Glucosinolates Mediated Regulation of Enzymatic Activity in Response to Oxidative Stress in Brassica spp.Plants (Basel). 2024 Dec 5;13(23):3422. doi: 10.3390/plants13233422. Plants (Basel). 2024. PMID: 39683215 Free PMC article. Review.
-
A new source of bacterial myrosinase isolated from endophytic Bacillus sp. NGB-B10, and its relevance in biological control activity.World J Microbiol Biotechnol. 2022 Sep 3;38(11):215. doi: 10.1007/s11274-022-03385-3. World J Microbiol Biotechnol. 2022. PMID: 36056962 Free PMC article.
-
Sulfate Availability and Hormonal Signaling in the Coordination of Plant Growth and Development.Int J Mol Sci. 2024 Apr 3;25(7):3978. doi: 10.3390/ijms25073978. Int J Mol Sci. 2024. PMID: 38612787 Free PMC article. Review.
-
Phylum Level Diversity of Plant Interior Bacteria in Seeds, Supernatant and Pellet Phases of Seed Suspension of Mustard Plant.Indian J Microbiol. 2024 Dec;64(4):1587-1597. doi: 10.1007/s12088-023-01184-4. Epub 2024 Feb 10. Indian J Microbiol. 2024. PMID: 39678952
-
The Effects of Interventions with Glucosinolates and Their Metabolites in Cruciferous Vegetables on Inflammatory Bowel Disease: A Review.Foods. 2024 Nov 1;13(21):3507. doi: 10.3390/foods13213507. Foods. 2024. PMID: 39517291 Free PMC article. Review.
References
-
- Yactayo-Chang J.P., Tang H.V., Mendoza J., Christensen S.A., Block A.K. Plant Defense Chemicals against Insect Pests. Agronomy. 2020;10:1156. doi: 10.3390/agronomy10081156. - DOI
-
- Del Castillo B., Garcia de Marina A., Martinez-Honduvilla M.P. Fluorimetric determination of oenanthotoxin. Ital. J. Biochem. 1980;29:233–237. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

