Anti-HIV Activity of Snake Venom Phospholipase A2s: Updates for New Enzymes and Different Virus Strains
- PMID: 35163532
- PMCID: PMC8835987
- DOI: 10.3390/ijms23031610
Anti-HIV Activity of Snake Venom Phospholipase A2s: Updates for New Enzymes and Different Virus Strains
Abstract
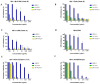
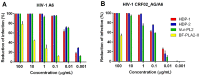
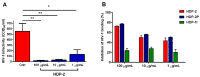
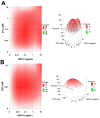
Since the beginning of the HIV epidemic, lasting more than 30 years, the main goal of scientists was to develop effective methods for the prevention and treatment of HIV infection. Modern medicines have reduced the death rate from AIDS by 80%. However, they still have side effects and are very expensive, dictating the need to search for new drugs. Earlier, it was shown that phospholipases A2 (PLA2s) from bee and snake venoms block HIV replication, the effect being independent on catalytic PLA2 activity. However, the antiviral activity of human PLA2s against Lentiviruses depended on catalytic function and was mediated through the destruction of the viral membrane. To clarify the role of phospholipolytic activity in antiviral effects, we analyzed the anti-HIV activity of several snake PLA2s and found that the mechanisms of their antiviral activity were similar to that of mammalian PLA2. Our results indicate that snake PLA2s are capable of inhibiting syncytium formation between chronically HIV-infected cells and healthy CD4-positive cells and block HIV binding to cells. However, only dimeric PLA2s had pronounced virucidal and anti-HIV activity, which depended on their catalytic activity. The ability of snake PLA2s to inactivate the virus may provide an additional barrier to HIV infection. Thus, snake PLA2s might be considered as candidates for lead molecules in anti-HIV drug development.
Keywords: antiviral activity; human immunodeficiency virus; phospholipase A2; pseudovirus; recombinant form; retrovirus; snake venom; syncytium.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of the data; in the writing of the manuscript; or in the decision to publish the results.
Figures


References
-
- Keele B.F., Giorgi E.E., Salazar-Gonzalez J.F., Decker J.M., Pham K.T., Salazar M.G., Sun C., Grayson T., Wang S., Li H., et al. Identification and characterization of transmitted and early founder virus envelopes in primary HIV-1 infection. Proc. Natl. Acad. Sci. USA. 2008;105:7552–7557. doi: 10.1073/pnas.0802203105. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

